外科病理学实践:诊断过程的初学者指南 | 第25章 涎腺
第25章 涎腺(Salivary Gland)
背景(Background)
With the exception of the pleomorphic adenoma, salivary gland neoplasms are rare, so you will not see many during residency training. To make matters worse, there is a great deal of morphologic overlap in some of the tumors, and immunostains are not usually helpful in distinguishing them. Your goal, early in your training, should be to recognize the more classic forms of the major tumors and also to be able to create a short differential diagnosis for any given tumor. In this organ, with all the mimics and variants, it is extremely important to approach a specimen with the question, “What else could this be?”
除了多形性腺瘤,涎腺肿瘤罕见,所以在住院医师培训期间你们不会看到很多。更糟糕的是,有些肿瘤存在大量的形态学重叠,免疫染色通常无助于区分。在训练的早期阶段,你的目标应该是识别较典型的主要肿瘤,并且能够对任何特定的肿瘤列举一些鉴别诊断。在这个器官中,有着所有的假冒病变和变异型,处理一个病例时带着问题“这还能是什么?”是极其重要的。
Biopsies of the salivary gland are occasionally performed in search of Sjögren’s syndrome; this is a complex diagnosis with specific criteria that must be met (see your favorite pathology textbook for that). Inflammatory lesions can also create a mass, such as chronic sialadenitis or a lymphoepithelial cyst.
涎腺活检偶尔用于寻找干燥综合征;这是一个复杂的诊断,必须符合特定的标准(请参阅您最喜欢的病理学教科书)。炎症性病变也可形成肿块,如慢性涎腺腺炎或淋巴上皮囊肿。
解剖学(Anatomy)
There are three major and innumerable minor pairs of salivary glands. The largest, on the cheek, is the parotid, where most neoplasms arise. The smaller major glands are the sublingual and submandibular, under the tongue and jaw. In general, the smaller the gland, the higher the proportion of its neoplasms that are malignant. Salivary neoplasms can arise in virtually any part of the sinonasopharyngeal system.
有三对大涎腺的和无数小涎腺。腮腺最大,在面颊上,是最常见的肿瘤发生部位。舌下腺和下颌下腺较小,分别位于舌下和颌下。一般来说,腺体越小,其恶性肿瘤的比例就越高。肿瘤几乎可以发生在鼻咽系统的任何部位。
正常组织学(Normal Histology)
The first major cell type is the secretory cell. The salivary glands are composed of serous and mucinous secretory units and ducts (Figure 25.1). Serous cells are wedge shaped (like pie slices) and arranged in acini around ducts. They are full of blue to purple granules. Mucinous cells have basal nuclei and apical mucin, like goblet cells; these are also arranged in acinar formations. The parotid is primarily serous, the submandibular is mixed, and the sublingual is primarily mucinous.
第一种主要细胞类型是分泌细胞。涎腺由浆液性和粘液性分泌单位和导管组成(图25.1)。浆液细胞呈楔形(像饼状切片),排列在导管周围的腺泡中。它们充满蓝色到紫色的颗粒。粘液细胞核位于基底,粘液位于顶端,像杯状细胞;它们也排列成腺泡状。腮腺主要为浆液性,颌下腺为混合性,舌下腺主要为粘液性。
The second major cell type is the duct cell. The duct system has three types of ducts: the terminal, or intercalated ducts; the intermediate-sized striated ducts; and the interlobular large ducts. Each has a different epithelium and is theoretically associated with different tumor types. The intercalated ducts are small profiles with low cuboidal epithelium, similar to a bile ductule (Figure 25.2). Striated ducts are more proximal and are larger, with pink columnar cells full of mitochondria and striated basal borders (hard to see). Interlobular or excretory ducts have pseudostratified columnar epithelium with or without goblet and squamous metaplasia. Different tumors have some morphologic similarity to these different ducts, which may help you keep all of the neoplasms straight.
第二种主要细胞类型是导管细胞。导管系统有三种类型的导管:终末导管或闰管;中等大小的纹状管;小叶间大导管。每种导管都有不同的上皮,理论上与不同的肿瘤类型相关。闰管小,低立方上皮,类似于胆管(图25.2)。纹状管更靠近近端,更大,粉红色柱状细胞,充满线粒体,基底边缘有条纹(很难看到)。小叶间导管或排泄管有假复层柱状上皮,有或无杯状上皮和鳞状化生。不同的肿瘤与这些不同的导管具有形态学相似性,这可能帮助你容易理解所有肿瘤。
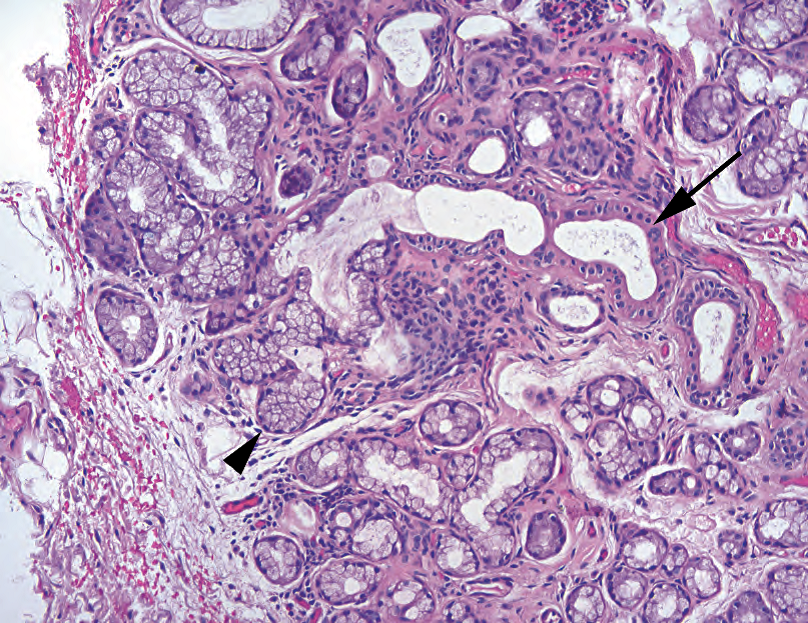
Figure 25.1. Normal salivary gland. In this example of mucinous salivary gland, the columnar secretory cells (arrowhead) form acini arranged around salivary ducts (arrow). Myoepithelial cells are not particularly visible on H&E stain.
图25.1 正常涎腺。这是粘液性涎腺,柱状分泌细胞(箭头)形成腺泡,排列在涎腺导管(箭)周围。肌上皮细胞在HE染色不是特别明显。
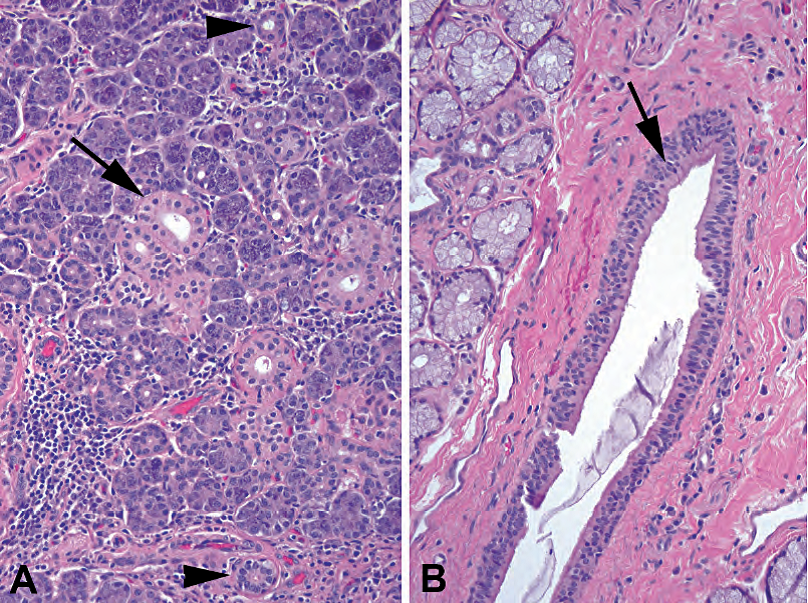
Figure 25.2. Types of ducts. (A) In the parotid, which has mainly serous glands, the terminal or intercalated ducts are visible as small tubules lined by cuboidal epithelium (arrowheads). The medium-sized striated ducts are more oncocytic in appearance, with abundant pink cytoplasm (arrow). (B) The large interlobular ducts have pseudostratified columnar epithelium (arrow), with occasional goblet cells, and become squamous at their junction with the gingival mucosa.
图25.2 导管类型。(A)腮腺主要是浆液腺,终末导管或闰管表现为被覆立方上皮的小管(箭头)。中等大小的纹状管更像嗜酸细胞,有丰富的粉红色细胞质(箭)。(B)大的小叶间导管有假复层柱状上皮(箭),偶有杯状细胞,在与牙龈粘膜连接处变为鳞状上皮。
The third major cell type is the myoepithelial cell. These cells, as in breast, surround acini and ducts. They are normally pale stellate cells with small nuclei and are very hard to identify in normal salivary gland. However, many neoplasms arise from the epithelial -myoepithelial cell line or, more specifically, from cells that can differentiate into either line. This creates a diagnostic nightmare, because the myoepithelial cells alone can take four different forms: spindled, plasmacytoid, epithelioid, or clear. Therefore, you must recognize any of these variants as myoepithelial (their immunologic profiles change with their form, unfortunately) and lump some very different-looking tumors into the same basket. Table 25.1 lists the most common neoplasms.
第三种主要细胞类型是肌上皮细胞。正如乳腺,这些细胞围绕腺泡和导管。正常情况下,它们是淡染的星状细胞,核小,在正常涎腺中很难识别。然而,许多肿瘤起源于上皮-肌上皮细胞系,或者更专业地说,来源于可分化为这两种细胞系的细胞。这就造成了一场诊断噩梦,因为肌上皮细胞本身可以有四种不同的形式:梭形、浆细胞样、上皮样或透明。因此,你必须识别这些变形的肌上皮(不幸的是,它们的免疫特征随其形态而变化),并将一些外观迥异的肿瘤归入同类。表25.1列出了最常见的肿瘤。
Table 25.1. Basic categories of the nine most common neoplasms.
表25.1 九种最常见肿瘤的基本分类。

Note that, in general, benign lesions are encapsulated, whereas malignant tumors are infiltrative, either as pushing fronts or as tendrils of cells (although mucoepidermoid carcinoma and acinic cell carcinoma can be deceptively well circumscribed). The first thing you should do when evaluating a salivary neoplasm is to study the periphery or capsule.
请注意,一般来说,良性病变有包膜,而恶性肿瘤呈浸润性,或表现为推挤性前沿,或表现为细胞的卷须(但粘液表皮样癌和腺泡细胞癌可能欺骗性地边界清楚)。当评估涎腺肿瘤时,你要做的第一件事是研究肿瘤周围或包膜。
肿瘤(Neoplasms)
This section describes the common neoplasms in order of how likely you are to see them, beginning with the most common.
本节从最常见的肿瘤开始,按照发生率顺序介绍常见肿瘤。
多形性腺瘤(良性混合瘤)(Pleomorphic Adenoma (Benign Mixed Tumor))
Pleomorphic adenoma is a biphasic tumor with epithelial and myoepithelial (mesenchymallike) components. It can occur anywhere but is very common in the parotid. The two key features to recognizing this lesion are a circumscribed, usually encapsulated tumor, and a mesenchymal-like component in the background (Figure 25.3). The mesenchymal-like stroma is often myxoid but may be chondroid or even osseous. The epithelial component can range from obviously epithelial to myoepithelial, so you may see well-defined ductular structures or pink to clear myoepithelial cells (in any of their four morphologies)—hence the designation pleomorphic. The individual cells, however, are notably not pleomorphic and in fact should be very bland (small, oval, pale nuclei).
多形性腺瘤是一种双相性肿瘤,具有上皮和肌上皮(间叶样)成分。它可以发生在任何部位的涎腺,但腮腺非常常见。有两个关键特征,可以识别这种病变:局限的,通常有包膜的肿瘤;背景中的间叶样成分(图25.3)。间叶样基质常为核分裂样,但也可能软骨样,甚至骨化。上皮成分的范围,从明显的上皮细胞到肌上皮细胞,因此你可能会看到完好的导管结构或粉粉红色到透明的肌上皮细胞(四种肌上皮形态之任一)——因此称为多形性。然而,很明显,单个细胞没有多形性,事实上细胞形态学非常温和(小、卵圆形、淡染核)。
The lesion not to miss is the polymorphous low-grade adenocarcinoma, which can look very similar but has an infiltrative periphery (see later). You should also examine the tumor for cytologically malignant cells, which may represent a carcinoma ex-pleomorphic adenoma (a pleomorphic adenoma gone bad).
不可忽视的病变是多形性低级别腺癌,其形态非常相似,但有浸润性边界(见下文)。你还要检查肿瘤中有无细胞学恶性,这可能代表癌在多形性腺瘤中(多形性腺瘤恶变)。
The myoepithelioma is one end of the pleomorphic adenoma spectrum, with very little mesenchymal component and no ductular differentiation. As with a pleomorphic adenoma, it should be encapsulated and circumscribed. Immunostains can help you here, as myoepitheliomas should be positive for S100, cytokeratin, glial fibrillary acidic protein, and actin. Basal cell adenoma is also analogous to a pleomorphic adenoma, with no mesenchymal component, but with a population of basaloid cells.
肌上皮瘤是多形性腺瘤形态学谱系的一端,间叶成分很少,没有导管分化。正如多形性腺瘤,肌上皮瘤应该边界清楚、有包膜。免疫染色可能有帮助,因为肌上皮瘤应表达S100、CK、GFAP和actin。基底细胞腺瘤也类似于多形性腺瘤,没有间质成分,但有大量基底样细胞。
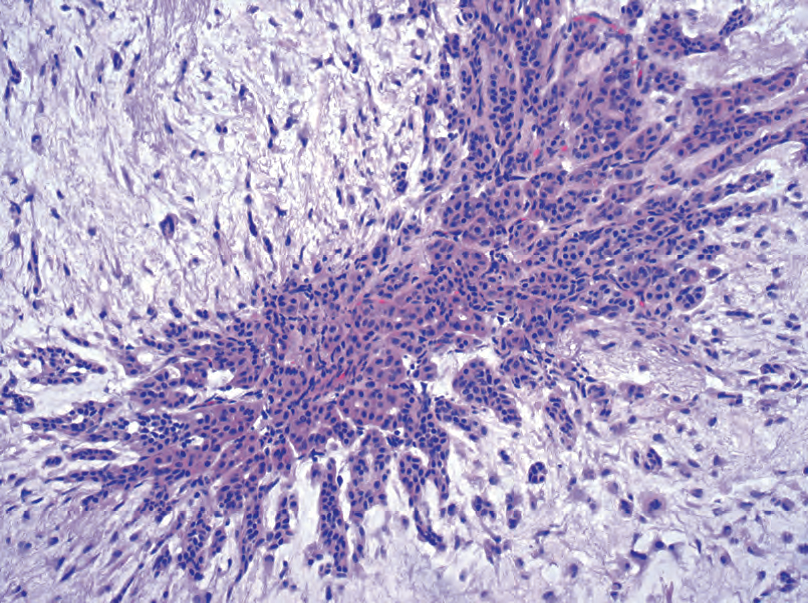
Figure 25.3. Pleomorphic adenoma. A cluster of cells is visible within the bluish myxoid stroma of a pleomorphic adenoma. The epithelial cells are small and cytologically benign, and they appear as small cords and tubules set within the stroma. The proportion of epithelial cells to stroma can vary widely.
图25.3 多形性腺瘤。蓝色核分裂样间质内可见一簇细胞。上皮细胞小,细胞学良性,在间质内呈小条索状和小管状。上皮细胞与基质的比例差异很大。
Rule of thumb: If it is an encapsulated solid lesion in the parotid, it probably fits into this category somehow.
经验法则:如果是腮腺内有包膜的实性病变,它可能在某种程度上属于这一类。
沃辛瘤(Warthin’s Tumor)
Warthin’s tumor is a papillary and cystic lesion lined by a double layer of oncocytic cells on top of a prominent lymphoid infiltrate with germinal centers (Figure 25.4). This usually occurs only in the parotid, but it can be bilateral. It is a low-power, 5-second diagnosis! This neoplasm (or reactive process?) may arise from striated ducts passing through intraparotid lymph nodes. The striated ducts are mitochondria rich, which explains the oncocytic nature of the lesion. This is to be differentiated from the lymphoepithelial cyst, a common lesion in patients with the HIV that has a thin ragged epithelial lining instead of an oncocytic one. A related oncocytic lesion is the oncocytoma—a lesion composed of oncocytes that looks similar to a Hürthle cell nodule in thyroid or an oncocytoma in the kidney.
Warthin瘤是一种乳头状囊性病变,被覆双层嗜酸细胞,下方为显著的淋巴浸润伴生发中心(图25.4)。通常只发生在腮腺,可能双侧发病。这是倍镜5秒诊断!这种肿瘤(或反应性过程?)可能起源于穿过腮腺内淋巴结的纹状管。纹状管富含线粒体,所以呈嗜酸性。这与淋巴上皮囊肿不同,后者是HIV患者的常见病变,其被覆上皮薄而不规则,而不是嗜酸细胞。相关的嗜酸细胞病变是嗜酸细胞瘤——一种由嗜酸细胞组成的病变,类似于甲状腺Hürthle细胞结节或肾脏嗜酸细胞瘤。
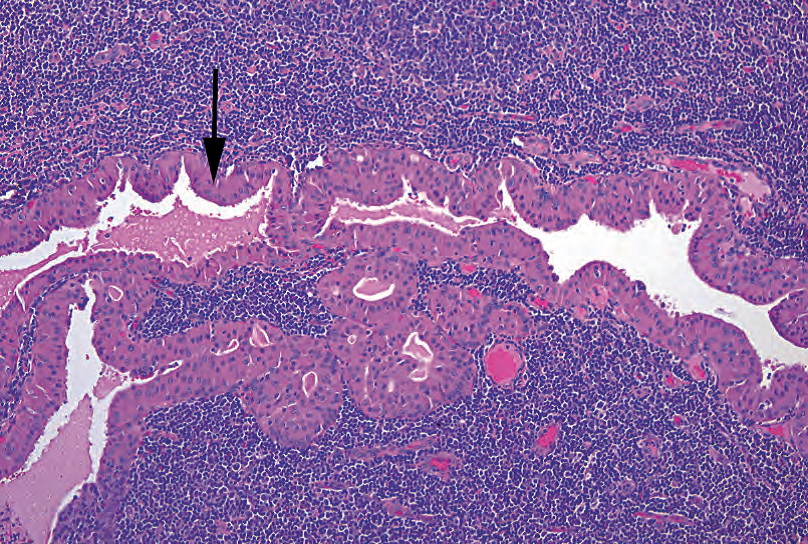
Figure 25.4. Warthin’s tumor. This cyst is lined by a double layer of oncocytic cells (arrow) overlying a dense lymphoid infiltrate.
图25.4 沃辛瘤。该囊肿被覆双层嗜酸细胞(箭),下方为密集的淋巴细胞浸润。
粘液表皮样癌(Mucoepidermoid Carcinoma)
Mucoepidermoid carcinoma is the most common malignant tumor. Mucoepidermoid carcinoma has a wide range of cellularity, from cystic to solid. It can be low grade to high grade, depending on five factors: percentage of cystic component, tumor necrosis, mitoses, anaplasia, and neural invasion. The neoplasm is a mixture of squamous cells, epithelioid cells, and clear mucinous cells. However, when one cell type dominates, it can be difficult to tell this tumor from (for example) squamous cell carcinoma or a clear cell carcinoma. In these cases, recognizing intracellular mucin is the key to the diagnosis, so a periodic acid-Schiff or mucicarmine stain may be used.
它是最常见的恶性肿瘤。它的细胞量变化很大,从囊性到实性。根据五个因素,把它分为低级别到高级别:囊性成分百分比、肿瘤坏死、核分裂、间变和神经浸润。肿瘤由鳞状细胞、上皮样细胞和透明的粘液细胞混合组成。然而,当一种细胞类型为主时,很难区分其他肿瘤(如,鳞状细胞癌或透明细胞癌)。在这些情况下,识别细胞内粘蛋白是诊断的关键,因此可以使用PAS或黏液卡红染色。
The periphery should be infiltrative, not encapsulated, although the low-grade tumors may be fairly well circumscribed. This tumor may arise from the interlobular ducts (the big excretory ducts), and you will notice that an inflamed or metaplastic duct does not look that different from a little focus of low-grade mucoepidermoid carcinoma (Figure 25.5).
肿瘤周围应该是浸润性,而不是包裹性(没有包膜),尽管低级别肿瘤的边界可能相当清楚。肿瘤可能起源于小叶间导管(大的排泄管),你会注意到炎症性或化生的导管与低级别粘液表皮样癌的小病灶没有什么不同(图25.5)。
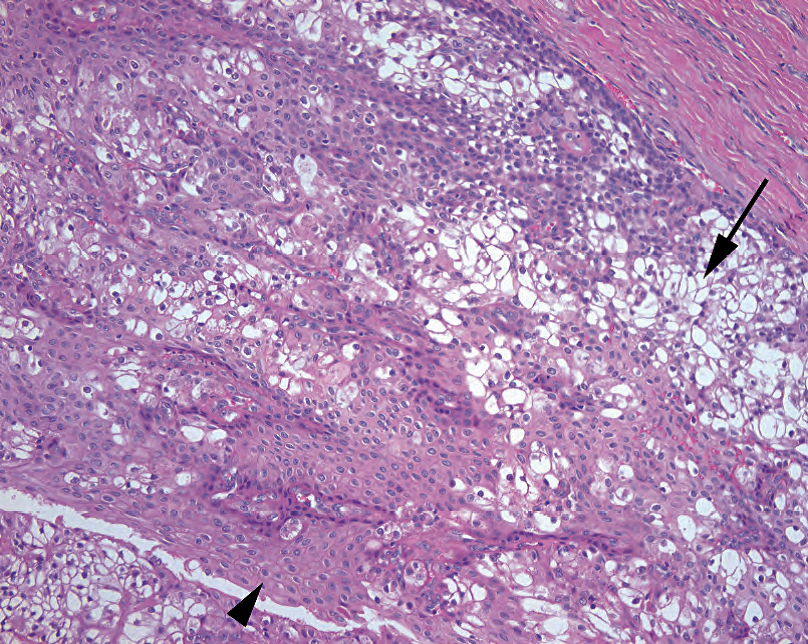
Figure 25.5. Mucoepidermoid carcinoma, low grade. This tumor resembles the metaplastic epithelium within an interlobular duct and is composed predominantly of clear goblet-like mucinous cells (arrow) and squamous cells (arrowhead).
图25.5 粘液表皮样癌,低级别。肿瘤类似于小叶间导管内的化生上皮,主要由透明的杯状粘液细胞(箭)和鳞状细胞(箭头)组成。
A low-grade cystic mucoepidermoid carcinoma must be distinguished from a mucin-containing salivary duct cyst, usually by carefully examining the cyst wall. As mentioned earlier, an intermediate-to-high grade mucoepidermoid carcinoma may be confused with metastatic or primary squamous cell carcinoma or clear cell carcinoma.
低级别囊性粘液表皮样癌必须区分含有粘液的涎腺导管囊肿,仔细检查囊壁通常可以鉴别。如前所述,中到高级别粘液表皮样癌可能与转移性或原发性鳞状细胞癌或透明细胞癌相混淆。
腺样囊性癌(Adenoid Cystic Carcinoma)
Adenoid cystic carcinoma is the prototypical cribriform tumor. It is blue, very cellular, with high nuclear to cytoplasmic ratios and fairly dense nuclei. Visually, it is reminiscent of a basal cell carcinoma of skin. It can be solid or tubular, but cribriform is the classic presentation, which makes it often instantly identifiable (Figure 25.6). Another classic feature is the balls of hyaline material found in the cribriform lumens, which are basement membrane material. It is highly infiltrative and loves nerves. Although intermediate grade at baseline, if solid growth predominates, this is a high-grade tumor.
腺样囊性癌是典型的筛状肿瘤。它是蓝色的,细胞非常丰富,具有高核质比和相当致密的细胞核。看起来像皮肤基底细胞癌。它可能呈实性或管状,但筛状是典型表现,使它一眼就能诊断(图25.6)。另一个典型特征是筛孔中的透明小球,这是基底膜物质。肿瘤侵袭性强,嗜神经。肿瘤至少是中级别,如果以实体生长为主,则为高级别。
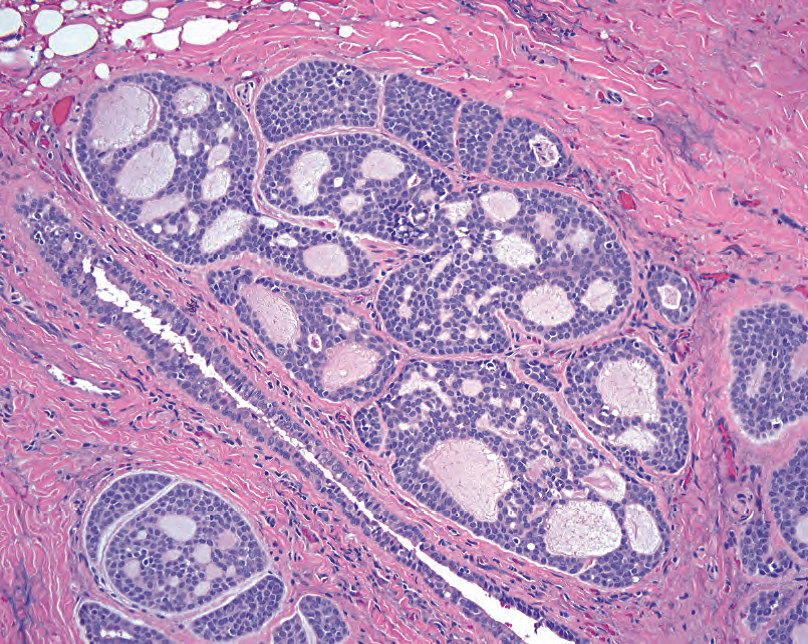
Figure 25.6. Adenoid cystic carcinoma. Although the nuclei are small, the nuclear to cytoplasmic ratio is high, making the tumor appear blue at low power. The architecture is classically cribriform, with sharply punched-out spaces full of pink secretions.
图25.6 腺样囊性癌。虽然核小,但核质比高,使肿瘤在低倍镜下呈蓝色。这是典型的筛状结构,有锐利的凿孔样腔隙,其中充满粉红色分泌物。
The presence of squamous areas favors a basaloid squamous cell carcinoma, which is in the differential diagnosis. The adenoid cystic carcinoma is very similar to the cylindroma of the skin, which can be in the differential around the ear (skin vs. tail of the parotid). Finally, if occurring in the lip or palate, rule out polymorphous low-grade adenocarcinoma, which can sometimes mimic an adenoid cystic. If you have a tumor that is cribriform and reminds you of an adenoid cystic, but is encapsulated or well-circumscribed, think instead of one of the adenomas, such as a basal cell adenoma.
存在鳞状区域倾向于基底样鳞状细胞癌,这是鉴别诊断之一。腺样囊性癌很像皮肤圆柱瘤,发生在耳朵周围时,需要鉴别(皮肤还是腮腺尾部?)。最后,如果发生在唇或腭部,要排除多形性低级别腺癌,它有时可能貌似腺样囊性病变。如果是筛状肿瘤,让你想起腺样囊性肿瘤,但有包膜或边界清楚,那就要考虑某种腺瘤,比如基底细胞腺瘤。
腺泡细胞癌(Acinic Cell Carcinoma)
Acinic cell carcinoma is a tumor of the serous acinar cells, and in its most differentiated form it looks quite similar to normal parotid, except without the ducts (Figure 25.7). Acinic cell is invasive, but as pushing borders rather than single cells. There are four common architectural patterns: solid, microcystic, papillary cystic, and follicular (such as thyroid). The microcystic pattern is full of little holes, but they are irregular holes that look as though the tissue has been pushed apart by expanding bubbles, very different from the rigid punched-out holes of adenoid cystic (Figure 25.8). There can be other nonserous cell types present, too, including clear cells, vacuolated cells, and ductal cells. However, finding a focus of serous differentiation (blue granular cytoplasm) pretty much seals the diagnosis.
腺泡细胞癌是浆液性腺泡细胞发生的肿瘤,其分化程度最高的形态很像正常腮腺,只是没有导管(图25.7)。它有浸润性,但不是单个细胞浸润,而是边缘呈推挤性浸润。有四种常见的结构模式:实性、微囊、乳头状囊和滤泡(如甲状腺)。微囊模式充满了小孔,但它们是不规则的孔,看起来像是组织被膨胀的气泡推开了,与腺样囊性癌的僵硬筛孔非常不同(图25.8)。也可能有其他非浆液性细胞类型,包括透明细胞、空泡细胞和导管细胞。然而,发现局灶浆液性分化(蓝色颗粒性细胞质)几乎可以确定诊断。

Figure 25.7. Acinic cell, solid pattern. The cells in acinic cell carcinoma replicate those of serous acini, with blue granular cytoplasm (arrow).
图25.7 腺泡细胞,实性模式。腺泡细胞癌中的细胞就像浆液性腺泡的细胞,胞质呈蓝色颗粒状(箭)。
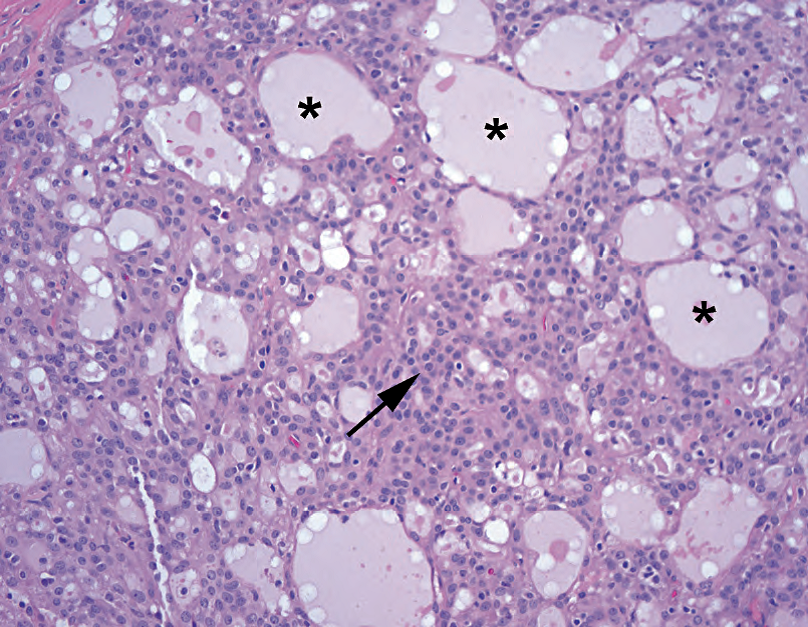
Figure 25.8. Acinic cell, microcystic pattern. In this example, the cells are nonspecifically pink (arrow) and do not have the telltale blue granules. However, the microcystic pattern, in which cells appear to be pushed apart by expanding pockets of fluid (asterisks), is typical of acinic cell. Compare these microcysts to the sharp cribriform spaces in adenoid cystic carcinoma (see Figure 25.6).
图25.8 腺泡细胞癌,微囊模式。在本例中,细胞呈非特异性粉红色(箭),没有蓝色颗粒。但有微囊模式,是腺泡细胞癌的典型表现,细胞似乎被扩张的液体囊(星号)推开。将这些微囊与腺样囊性癌的锐利筛孔进行比较(见图25.6)。
The nonserous tumors can be quite pink and nonspecific in appearance. However, look for the microcystic pattern, the absence of cytoplasmic mucin (rules out mucoepidermoid carcinoma), and uniform well-spaced nuclei (either small and dense or medium-sized and very round with small nucleoli), similar to the follicular thyroid lesions. Note the pronunciation: it is ass-inic, not ax-inic.
肿瘤中的非浆液性成分可能很红,外观也没有特异性。然而,寻找微囊模式、无细胞质黏液(排除粘液表皮样癌)和均匀分布的细胞核(小而致密,或中等大小、非常圆且有小核仁),类似于甲状腺滤泡性病变,有助于诊断腺泡细胞癌。注意拼写:它是ass inic,不是ax inic。
腺癌,非特指(NOS)(Adenocarcinoma Not Otherwise Specified (NOS))
Adenocarcinoma NOS is the category for any ductal-derived carcinoma without other distinguishing features. It is the most common form of malignancy in a carcinoma ex-pleomorphic adenoma, which is a pleomorphic adenoma with a component of carcinoma.
腺癌NOS是导管起源的癌,没有其他分类特征。它是癌在多形性腺瘤中最常见的恶性肿瘤,即,多形性腺瘤伴癌成分。
多形性低级别腺癌(Polymorphous Low-Grade Adenocarcinoma)
Importantly, polymorphous low-grade adenocarcinoma (PLGA) occurs almost exclusively in the intraoral minor salivary glands (lip and palate), so do not agonize over a funny-looking pleomorphic adenoma in the parotid! The PLGA looks very similar to a pleomorphic adenoma in terms of the epithelial component, and like a pleomorphic adenoma the cells are very bland. The key is in the margin of the tumor, which in a PLGA is infiltrative (Figure 25.9). The cells tend to spiral out of the central mass like a hurricane and may remind you of lobular carcinoma (its former name, incidentally). Polymorphous low-grade adenocarcinoma can also sometimes be cribriform in appearance, mimicking an adenoid cystic.
重要的是,多形性低级别腺癌(PLGA)几乎只发生在口腔内的小涎腺(唇和腭),所以不要为腮腺中一个看起来滑稽的多形性腺瘤而苦恼!就上皮成分而言,PLGA看起来很像多形性腺瘤,并且,正如多形性腺瘤,细胞非常温和。关键是肿瘤边缘,PLGA呈浸润性的边缘(图25.9)。这些细胞倾向于像飓风一样从中心肿块螺旋出来,可能会让你想起小叶癌(顺便说一句,它以前的名字)。PLGA有时呈筛状,类似于腺样囊性癌。
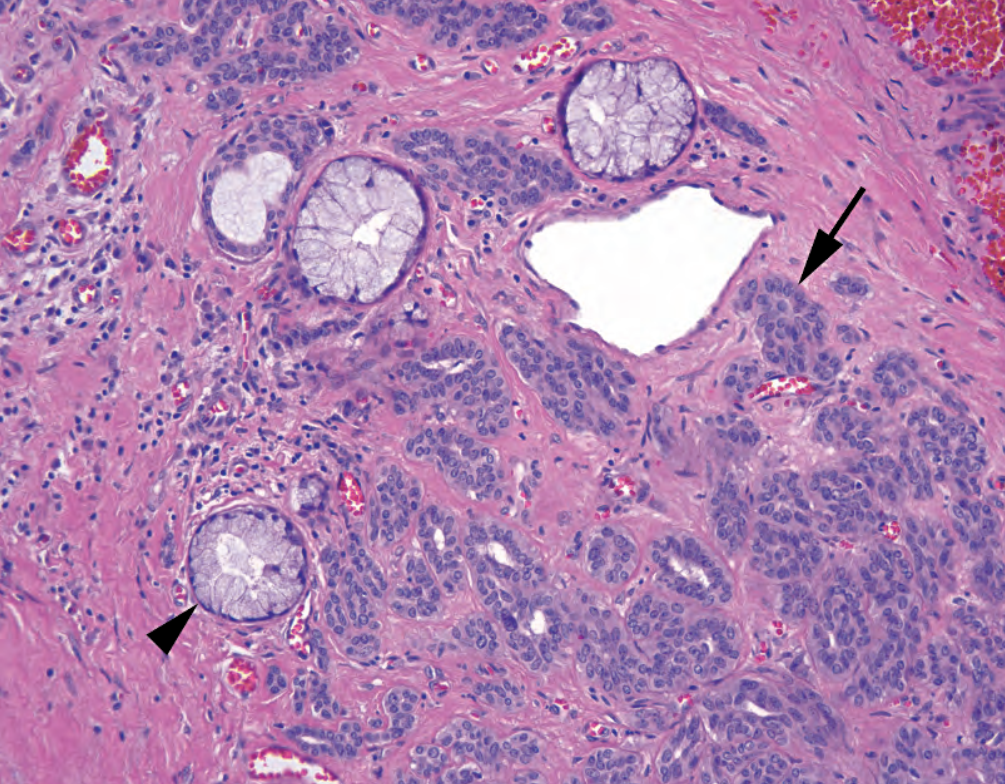
Figure 25.9. Polymorphous low-grade adenocarcinoma. Small tubules of bland cells (arrow) creep between benign mucinous glands (arrowhead). On high power, these infiltrative cells resemble those of the pleomorphic adenoma, but, unlike that benign tumor, the PLGA infiltrates surrounding tissues. PLGA cells may also invade as single-file lines, like lobular breast carcinoma.
图25.9 多形性低级别腺癌。温和细胞形成小管(箭),在良性粘液腺(箭头)之间爬行。高倍镜下,这些浸润性细胞像是多形性腺瘤,但与良性肿瘤不同,PLGA浸润周围组织。PLGA细胞也可以像乳腺小叶癌一样呈单行线状浸润。
杂项恶性肿瘤(Miscellaneous Malignant Neoplasms)
Many of the benign adenomas described here have malignant counterparts, although they are rare. They include myoepithelial carcinoma, epithelial–myoepithelial carcinoma, basal cell adenocarcinoma, and oncocytic carcinoma. In general, features that favor malignancy include an infiltrative periphery, cellular pleomorphism, mitoses, or necrosis.
这里描述的许多良性腺瘤都有对应的恶性肿瘤,尽管罕见。包括肌上皮癌、上皮-肌上皮癌、基底细胞腺癌和嗜酸细胞癌。一般来说,倾向于恶性肿瘤的特征包括浸润性边缘、细胞多形性、核分裂或坏死。
来源:
The Practice of Surgical Pathology:A Beginner’s Guide to the Diagnostic Process
外科病理学实践:诊断过程的初学者指南
Diana Weedman Molavi, MD, PhD
Sinai Hospital, Baltimore, Maryland
ISBN: 978-0-387-74485-8 e-ISBN: 978-0-387-74486-5
Library of Congress Control Number: 2007932936
© 2008 Springer Science+Business Media, LLC
仅供学习交流,不得用于其他任何途径。如有侵权,请联系删除
本站欢迎原创文章投稿,来稿一经采用稿酬从优,投稿邮箱tougao@ipathology.com.cn
相关阅读
 数据加载中
数据加载中
我要评论

热点导读
-

淋巴瘤诊断中CD30检测那些事(五)
强子 华夏病理2022-06-02 -
【以例学病】肺结节状淋巴组织增生
华夏病理 华夏病理2022-05-31 -

这不是演习-一例穿刺活检的艰难诊断路
强子 华夏病理2022-05-26 -

黏液性血性胸水一例技术处理及诊断经验分享
华夏病理 华夏病理2022-05-25 -

中老年女性,怎么突发喘气困难?低度恶性纤维/肌纤维母细胞性肉瘤一例
华夏病理 华夏病理2022-05-07







共0条评论