外科病理学实践:诊断过程的初学者指南 | 第11章 前列腺
第11章 前列腺(Prostate)
Most biopsies are performed for an elevated prostate-specific antigen (PSA) level, a palpable nodule, or a history of an abnormal biopsy. In the prostate, you are generally looking only for adenocarcinoma; there are very few nonneoplastic conditions to look for.
大多数活检是针对前列腺特异性抗原(PSA)水平升高、可触及结节或异常活检史。在前列腺,通常只要寻找腺癌;非肿瘤性疾病很少。
A typical sextant biopsy is six cores from left apex, mid, and base and right apex, mid, and base. Increasingly, urology centers are sampling additional areas, 12 or more. Laboratories differ in how many cores are placed on a single slide; some laboratories may have only two slides, left and right, with a handful of cores on each slide. It is important to preserve as much detail as the urologist or laboratory gives you and to localize the cancer as much as possible.
典型的6点活检是从左侧的尖、中、底和右侧的尖、中、底共取6根穿刺组织条。越来越多的泌尿中心正在增加取样,共取12点或更多区域。实验室在一张切片上放置的组织条数不同;一些实验室可能只有左右两张切片,每张切片上都有一些的穿刺组织条。重要的是尽可能多地保留泌尿科医生或实验室给你的细节,并尽可能定位癌。
粗针穿刺活检的观察方法(Approach to the Core Biopsy)
On 4× to 10×, scan the length of the core looking for glands that stand out and look different.
在4×到10×镜下,扫描穿刺组织条的整个长度,寻找引人注目的、与众不同的腺体。
Low-power features of prostate cancer (Figure 11.1)
前列腺癌的低倍特征(图11.1)
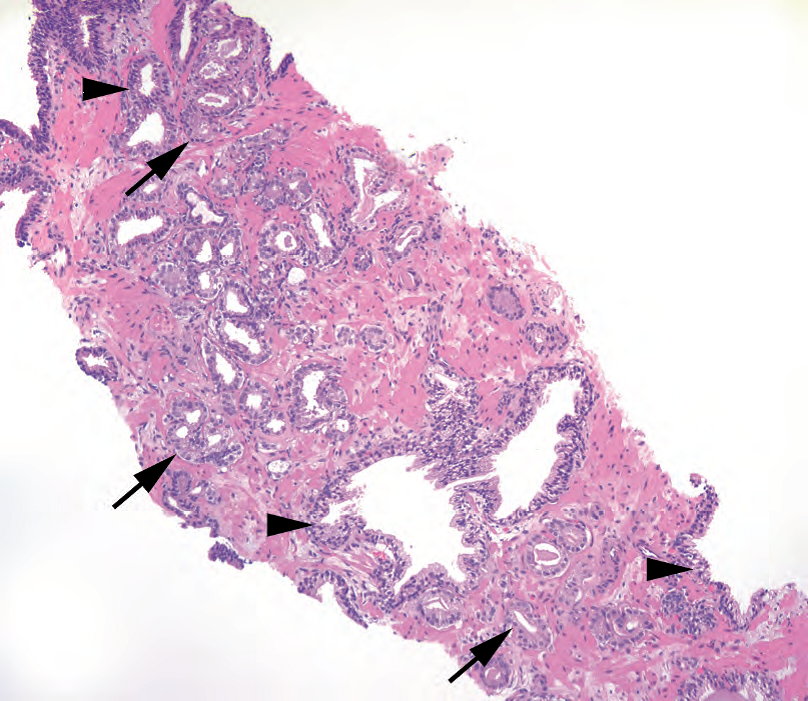
Figure 11.1. Low-power features of carcinoma. Adenocarcinoma (arrows) is seen infiltrating throughout benign glands (arrowheads) in this core biopsy specimen. The malignant glands are often back to back and have relatively denser cytoplasm, no basal layer, and straight luminal borders.
图11.1.癌的低倍特征。在这个粗针穿刺活检标本中,可以看到腺癌(箭)浸润于整个良性腺体(箭头)之间。恶性腺体通常背靠背,细胞质相对深染,无基底层,管腔边界笔直。
° Small individual glands infiltrating among larger benign glands (for intermediate-grade cancer, or Gleason pattern 3; see more on Gleason grades, discussed later)
° 在较大的良性腺体之间浸润的单个小腺体(适用于中间级别癌,或Gleason模式3;下文讨论更多的Gleason分级)
° Crowded glands (for intermediate-grade cancer)
° 腺体拥挤(适用于中间级别癌)
° An unusually cellular infiltrate (individual cells of high-grade cancer, or Gleason pattern 5)
° 不同寻常的细胞浸润(高级别癌的单个细胞,或Gleason模式5)
° Cribriform areas (high-grade cancer)
° 筛状区域(高级别癌)
° Sheets of cells (high-grade cancer)
° 细胞成片(高级别癌)
° A different color or texture to the glands (cancer may appear denser or more bluish)
° 腺体的颜色或质地不同(癌可能更深染或更蓝)
° Blue mucin, crystalloids, or dense pink secretions in the lumen
° 管腔内有蓝色黏液、晶体或深染的粉红色分泌物
° Absence of desmoplastic response
° 无促结缔组织增生反应
Low-power features of benign glands (Figure 11.2)
良性腺体的低倍特征(图11.2)
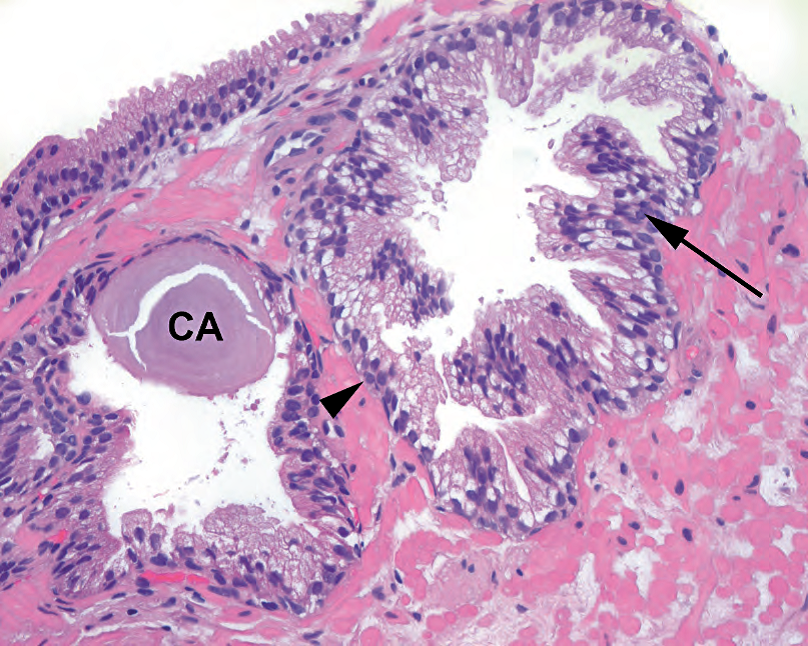
Figure 11.2. Benign prostate glands. These glands have a distinct basal cell layer underlying the epithelial cells (arrowhead) and papillary fronds in the lumen (arrow). Corpora amylacea (CA) are concentrically laminated concretions associated with benign glands.
图11.2.良性前列腺。这些腺体在上皮细胞(箭头)下方和管腔中的乳头状突起(箭)下方有一层明显的基底细胞层。淀粉样小体(CA)是与良性腺体相关的同心层状结石。
° Irregularly shaped glands with papillary infoldings (a “frilly” look)
° 形状不规则的腺体,有乳头状内折(“褶边状”)
° Glands with a modest amount of intervening stroma
° 腺体之间有少量间质
° Corpora amylacea
° 淀粉样小体
At high power (20×–40×), examine the cytology of the suspicious areas. Look for features seen in carcinoma:
在高倍镜下(20×–40×),检查可疑区域的细胞学。寻找癌的特征:
° Large, often cherry-red nucleoli (Figure 11.3)
° 大的,通常是樱桃红色的核仁(图11.3)
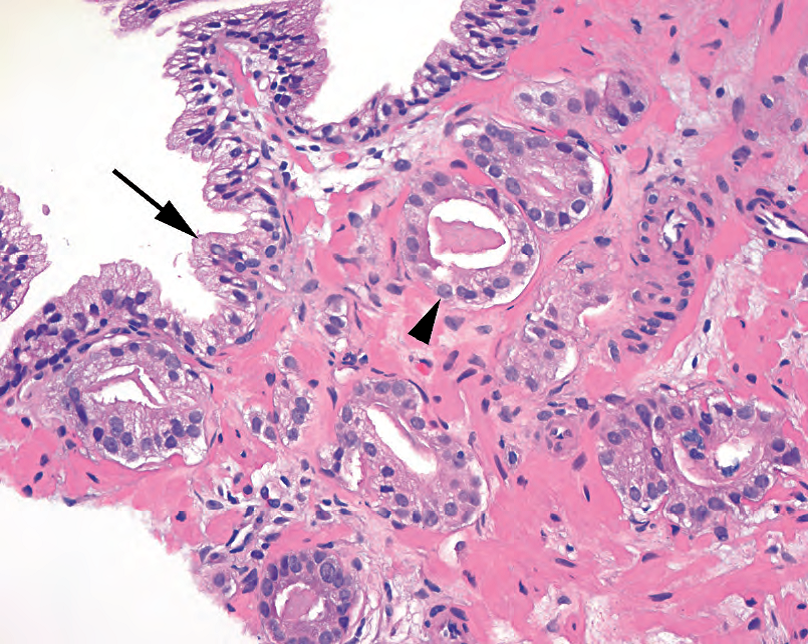
Figure 11.3. High-power features of carcinoma. Malignant glands show distinct nucleoli (arrowhead), sharp luminal borders, and an absence of basal cells. Benign glands are seen adjacent to the cancer(arrow).
图11.3.癌的高倍特征。恶性腺体显示明显的核仁(箭头),管腔边缘锐利,基底细胞缺失。癌旁可见良性腺体(箭)。
° Straight, crisp luminal borders to the glands
° 腺体的管腔边界笔直、清晰
° Enlarged and/or hyperchromatic nuclei (however, pleomorphism is minimal)
° 核增大和/或核深染(但多形性轻微)
° Lack of basal cell layer (can be confirmed by immunostains)
° 缺乏基底细胞层(可通过免疫染色确认)
° Mitoses (uncommon)
° 核分裂(少见)
Although none of these findings is completely sensitive and specific for cancer, having more malignant than benign features is a pretty good indication. There are three features that, although uncommon, are only seen in cancer:
虽然这些发现对癌都不是完全敏感和特异的,但恶性特征多于良性特征是很好的提示。有3个特征虽然少见,但仅见于癌:
° Perineural invasion: The nerve appears as a discrete oval profile with wavy parallel stripes, almost like a fingerprint, and the malignant gland must be within the nerve sheath to count as perineural invasion (Figure 11.4). Often the gland will fill up the nerve sheath circumferentially, so the nerve appears to be floating in a gland.
° 神经周围侵犯:神经呈离散的椭圆形轮廓,有波浪状平行条纹,几乎像指纹。恶性腺体必须位于神经鞘内才算神经周围侵犯(图11.4)。通常腺体会填满神经鞘周围,因此神经似乎漂浮在腺体中。
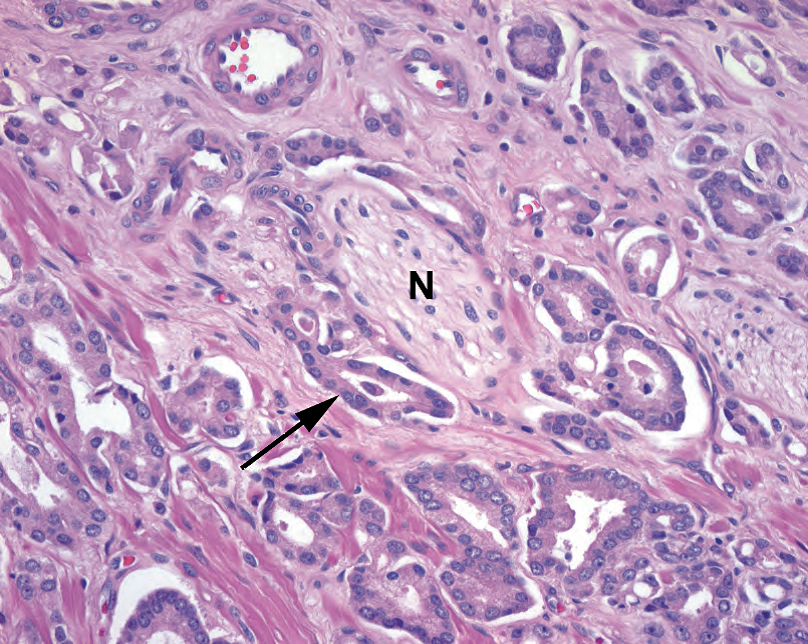
Figure 11.4. Perineural invasion. A nerve (N) is identified by the undulating axons and nerve sheath nuclei. Malignant glands are seen nearly surrounding the nerve (arrow).
图11.4.神经周围侵犯。神经(N)由起伏的轴突和神经鞘核识别。神经周围可见恶性腺体(箭)。
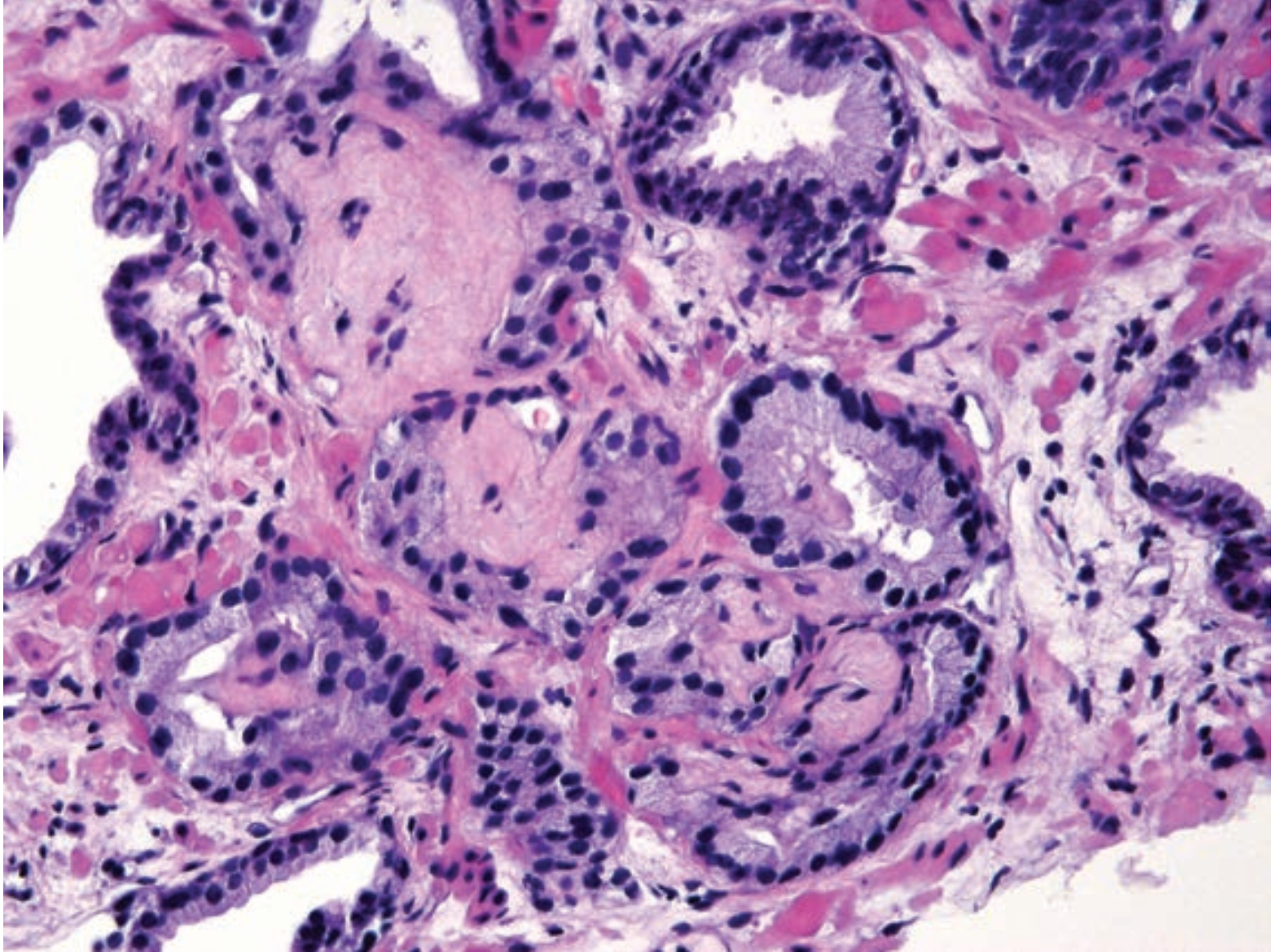
° Mucinous fibroplasia: Hyalinized whorls of organized dense secretions are present in the lumen; sometimes the surrounding gland epithelium may be compressed and indistinct.
° 黏液性纤维增生:管腔内出现似乎有组织结构的致密分泌物并有纤维母细胞长入,形成透明旋涡状结构;有时,周围的腺体上皮可能受压,模糊不清。
(译注:补充图片示前列腺腺癌中的黏液性纤维增生)
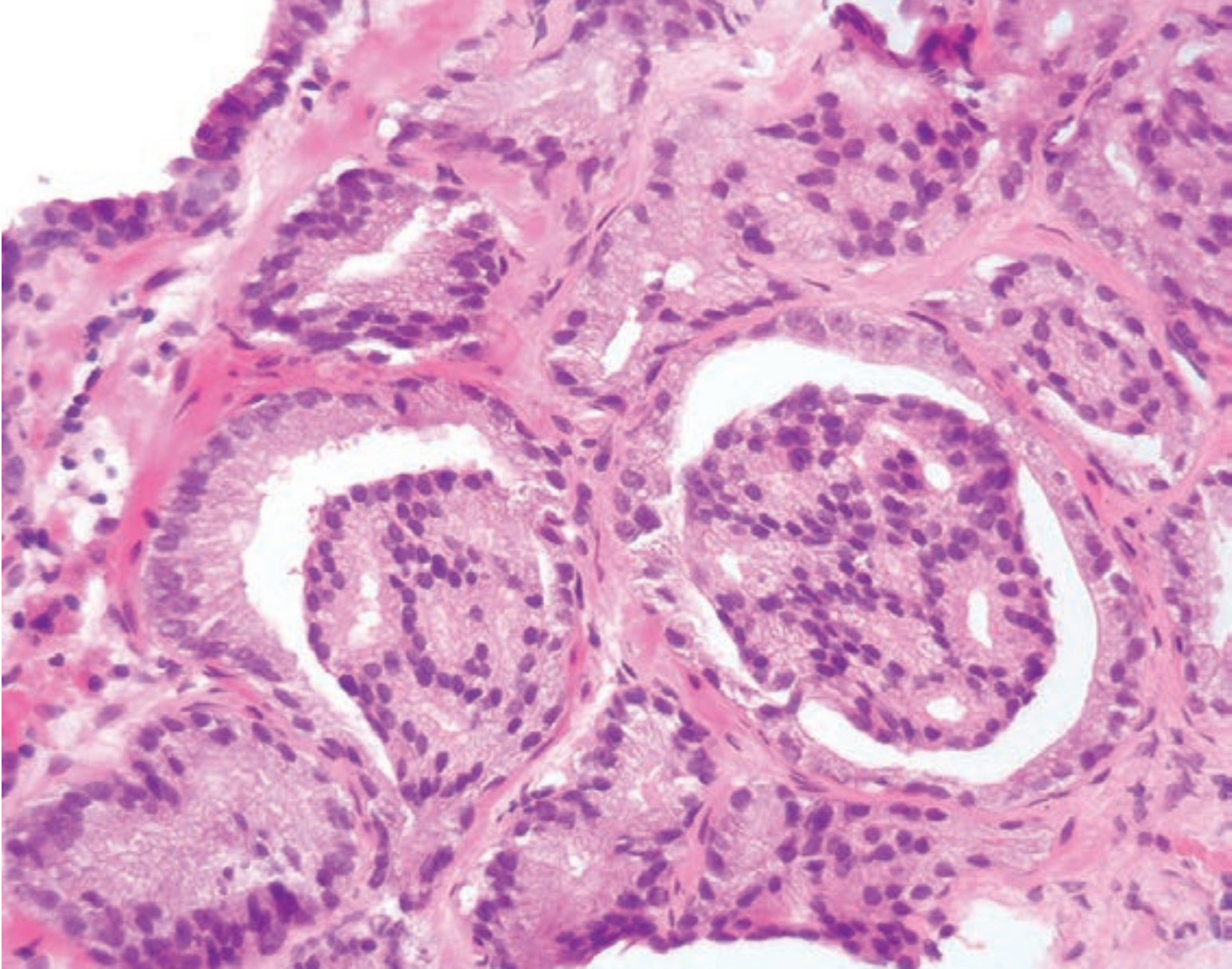
° Glomeruloid forms: Proliferative tangles of cells project into the larger gland lumen, resembling a glomerulus.
° 肾小球样结构:增殖的细胞缠结伸入较大的腺腔,像肾小球。
(译注:补充图片示前列腺腺癌中的肾小球样结构)
Gleason分级(Gleason Grading)
Once you have identified adenocarcinoma, you must give it a histologic grade. Prostatic adenocarcinoma is graded by the Gleason system, which is based on architectural pattern. Cytology does not affect grade. The patterns (and there may be many within a single tumor) are assigned a number from 1 to 5, with 5 being the least differentiated. After all tissue is examined, the first and second most common patterns are added together to give the Gleason score (a possible 2–10). A pure tumor of pattern 3 would be a 3 + 3 = 6; a mixture of 3 and 4 could be signed out as 4 + 3 = 7 or 3 + 4 = 7, depending on the amount of each.
确定腺癌后,需要组织学分级。Gleason分级系统基于组织学结构模式,而不是细胞学。这些结构模式(单个肿瘤内可能有多种结构模式)赋值1到5,其中5是分化最差的。检查所有组织后,将第一和第二最常见的模式相加,得出Gleason分数(可能为2-10)。模式3的纯肿瘤为3+3=6;3和4的混合物可以是4+3=7或3+4=7,具体取决于每种混合物的数量。
You may want to take the Johns Hopkins online tutorial for prostate grading, available at http:// pathology2.jhu.edu/gleason/. Other good grading websites can be found at www.isuporg.org. In summary, the pattern grades are as follows:
可以参加约翰•霍普金斯大学前列腺分级在线教程http://pathology2.jhu.edu/gleason/。其他优秀的评分网站可以在www.isuporg.org上找到。综上所述,模式级别如下:
1:rarely used; a circumscribed nodule of uniform crowded glands
1:很少使用;均匀一致的拥挤的腺体形成边界清楚的结节
2:a circumscribed nodule of well-defined glands with minimal infiltration at the periphery; less uniform than pattern 1.
2:形态完好的腺体,形成一个边界清楚的结节,外围有很少浸润;不如模式1那样均匀。
3:highly infiltrative (creeping between benign glands), with discrete and individual gland profiles such that you can mentally draw a circle around each gland (Figure 11.5).
3:高度浸润性(在良性腺体之间爬行),有独立的单个腺体轮廓,因此可以在每个腺体周围画一个圆圈(图11.5)。
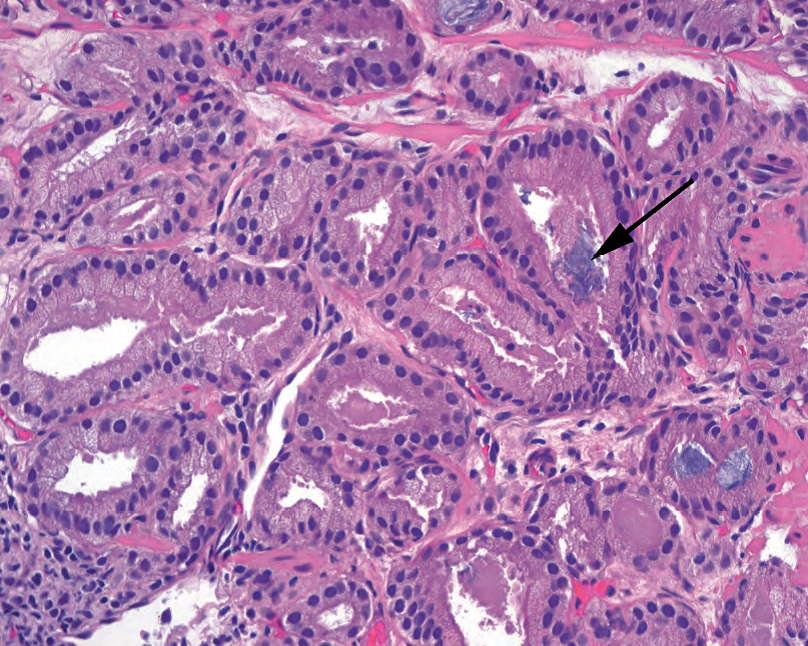
Figure 11.5. Gleason pattern 3. Individual, well-formed malignant glands make up pattern 3 cancer. Blue mucin, often associated with carcinoma, is present (arrow).
图11.5.Gleason模式3,由单个、形态完好的恶性腺体构成。出现蓝色黏液,通常与癌有关(箭)。
4:fused and ill-defined glands, sheets of cribriform glands, poorly formed lumens (Figure 11.6).
4:融合的、界限不清的腺体,成片的筛状腺体,形态不完好的管腔(图11.6)。
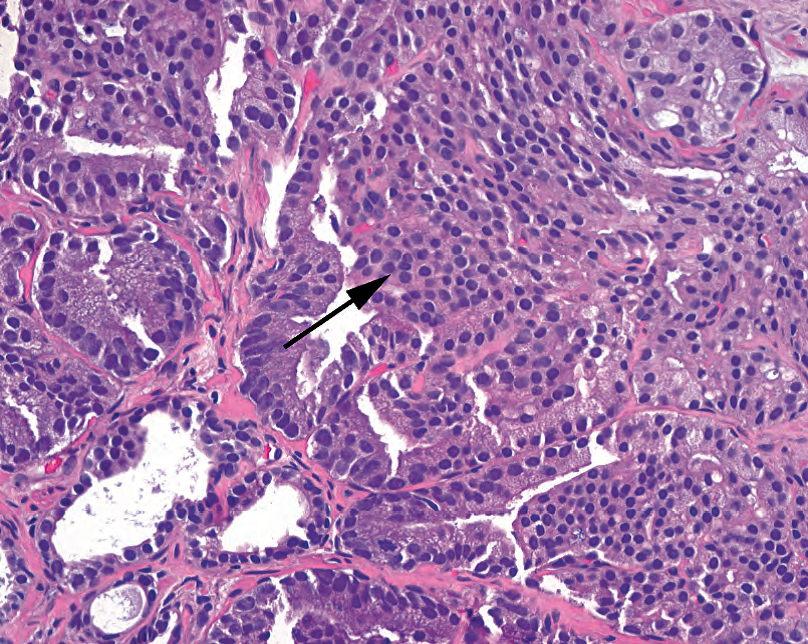
Figure 11.6. Gleason pattern 4. The area of cribriform growth (arrow) and adjacent fused glands is typical of pattern 4.
图11.6.Gleason模式4。筛状生长区域(箭)和相邻的融合腺体是模式4的典型表现。
5:a complete absence of glandular differentiation, solid sheets and cords of cells, single cells (Figure 11.7)
5:完全没有腺体分化,成片实性和条索状细胞,单个细胞(图11.7)
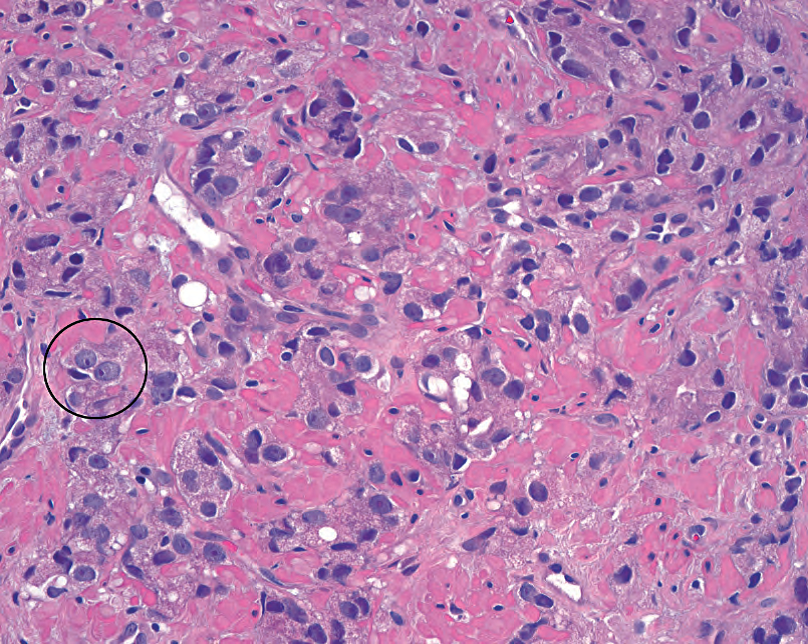
Figure 11.7. Gleason pattern 5. Individual malignant cells, without evidence of gland formation, are typical of pattern 5. The individual cells still cytologically resemble well-differentiated carcinoma, with round nuclei and prominent nucleoli (circle).
图11.7.Gleason模式5。单个恶性细胞,没有腺体形成证据,是模式5的典型表现。单个细胞在细胞学上仍然类似于高分化癌,具有圆核和显著的核仁(圆圈)。
Gleason scores of 2–4 (i.e., a combination of patterns 1 and 2) are not diagnosed on needle biopsy, as they can only be identified in the context of surrounding tissue.
Gleason评分为2–4(即模式1和模式2的组合)的不能在粗针穿刺活检中进行诊断,因为只能在周围组织的背景中进行识别。
诊断中应提及的特征(Features That Should Be Mentioned in Your Diagnosis)
The following features should be mentioned in your diagnosis:
在诊断中应提及以下特征:
Number of involved cores: A biopsy diagnosis should include a mention of how many cores are involved (e.g., 1 of 1 core, or 2 of 4 cores).
累及的穿刺组织条数量:活检诊断应包括累及的穿刺组织条数量(例如,1个穿刺组织条中的1个,或4个穿刺组织条中的2个)。
Percent involvement: Note the approximate percentage involvement on each core (e.g., involving 2 of 4 cores [30%, 60%]). Small foci of cancer (< 5% of a core) can be described as small foci.
累及百分比:注明每个穿刺组织条的累及百分比(例如,累及4个穿刺组织条中的2个[30%,60%])。癌的小病灶(<5%的穿刺组织条)可以描述为小灶。
Perineural invasion: Once cancer is identified, look closely for foci of perineural invasion. The presence of perineural invasion in a biopsy specimen has adverse prognostic significance.
神经周围浸润:一旦发现癌,仔细寻找神经周围浸润的病灶。活检标本中神经周围浸润是不利预后因素。
Extraprostatic extension: Rarely, a core biopsy will go through the capsule of the prostate and into the fat beyond. An extremely lucky shot may show malignant glands trickling into the fat, which is diagnostic of extraprostatic extension. This also has adverse prognostic significance.
前列腺外延伸:很少,粗针穿刺活检会穿过前列腺被膜,进入前列腺外脂肪。非常巧合的情况下,能捕捉到恶性腺体侵入脂肪,这是前列腺外延伸的诊断。这也是不利的预后因素。
前列腺上皮内肿瘤(Prostatic Intraepithelial Neoplasia)
Prostatic intraepithelial neoplasia (PIN) occupies a slightly uncertain place in pathology. It is considered to be a precursor to cancer and to demonstrate a generally increased risk of cancer, but, unlike precursor lesions such as high-grade squamous intraepithelial lesions (cervix) or ductal carcinoma in situ (breast), it does not warrant an immediate rebiopsy or excision. You can think of it as dysplasia in the prostate gland, but the natural history of the lesion is unclear. In any case, the lack of reproducibility and questionable significance of low-grade PIN are such that we do not mention it on biopsy. High-grade PIN, however, should be noted. Features of high-grade PIN include the following:
前列腺上皮内肿瘤(PIN)在病理学中占有一个稍微不确定的位置。它是癌的前驱病变,患癌风险总体上增加,但与高级鳞状上皮内病变(宫颈)或导管原位癌(乳腺)等前驱病变不同,它不促使临床立即进行再次活检或切除。你可以认为它是前列腺的异型增生,但病变的自然史尚不清楚。在任何情况下,低级别PIN缺乏重复性和可疑意义,因此我们在活检中不用提及。但是,应注意高级别PIN。高级别PIN的特点包括:
Glands are large with prominent papillary or micropapillary luminal surfaces, similar to benign architecture. Cribriform PIN can be seen, but back-to-back glands are not PIN.
腺体较大,有显著的乳头状或微乳头状腔面,类似于良性结构。可以看到筛状的针,但背靠背的腺体不是针。
Glands appear darker and more blue than surrounding glands (Figure 11.8).
腺体比周围的腺体更深染、更蓝(图11.8)。
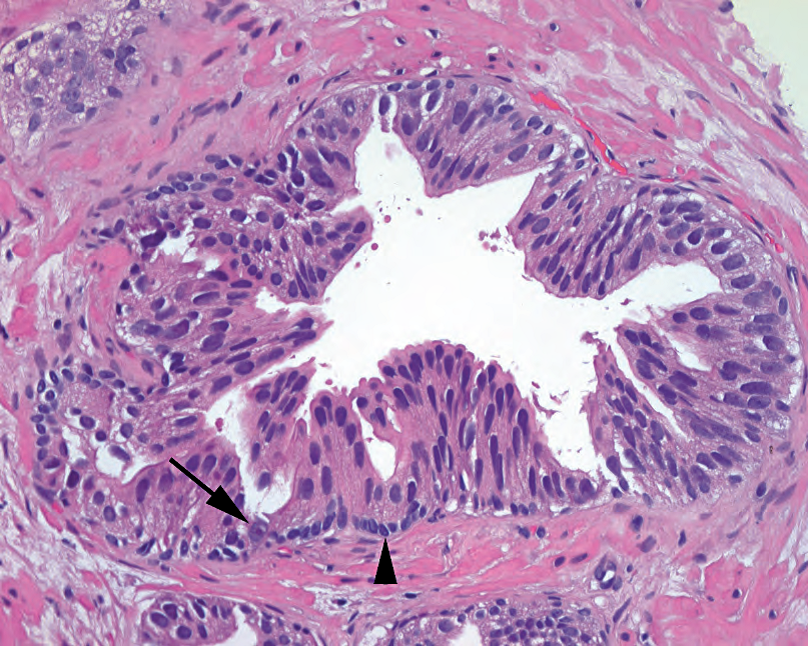
Figure 11.8. High-grade prostatic intraepithelial neoplasia. Although the papillary infoldings resemble benign prostate, the nuclei are larger and darker and show occasional prominent nucleoli (arrow). Basal cells are still present (arrowhead).
图11.8.高级别前列腺上皮内肿瘤。尽管乳头状内折类似于良性前列腺,但核更大更深染,偶见明显的核仁(箭)。基底细胞仍然存在(箭头)。
Nuclei are enlarged, elongated, and hyperchromatic, and by definition nucleoli are visible at 20×.
核增大、拉长、深染,根据定义,20x可见核仁。
The basal cell layer is usually still present, yet often patchy; immunostains show this nicely.
基底细胞层通常仍然存在,但通常是斑片状的;免疫染色很好地显示了这一点。
前列腺癌的假冒病变(Mimickers of Prostate Cancer)
There are some benign entities that may catch your eye and stand out in a biopsy specimen but that are definitely not carcinoma.
有一些良性实体可能会吸引你的眼球,并在活检标本中与众不同,但绝对不是癌。
腺病(Adenosis)
Adenosis literally means a proliferation of glands. Adenosis is a hyperplastic lesion, not a neoplastic one. It consists of a lobular group of crowded glands, which may include small suspicious-looking glands among them. The morphology of the small glands, however, should overlap with the intermixed larger, benign-looking glands; there should be a spectrum so that you cannot point to definite malignant versus benign glands. Adenosis may have visible small nucleoli (how unfortunate) but by definition has a basal layer (visible by immunostains if not by HE).
腺病的字面意思是指腺体增生。腺病是一种增生性病变,不是肿瘤性病变。它由拥挤的腺体形成小叶状聚集,其中可能包括外观可疑的小腺体。然而,小腺体的形态应该与混杂的较大的良性腺体相重叠;应该有一个连续的形态学谱系,可疑的恶性腺体与良性腺体无法区分。腺病可有小核仁(多么不幸),但根据定义,必需存在基底层(如果HE不清楚,则做免疫染色)。
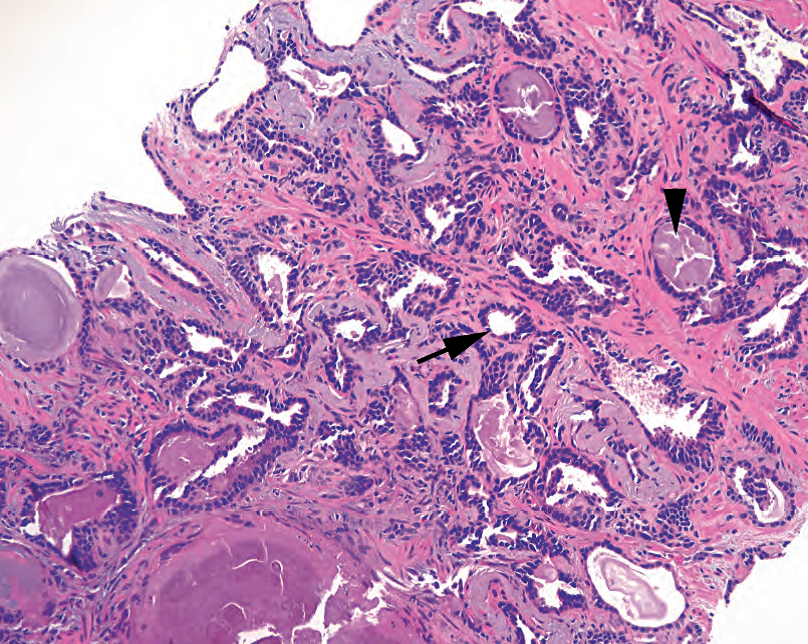
Figure 11.9. Atrophy. These glands appear hyperchromatic and infiltrative. However, the low cuboidal cells with attenuated cytoplasm (arrow) and angular gland profiles are typical of benign atrophy. Corpora amylacea are present (arrowhead).
图11.9.萎缩。这些腺体似乎深染和浸润。然而,低立方形细胞的胞质变少(箭),腺体成角,是良性萎缩的典型表现。存在淀粉样小体(箭头)。
萎缩(Atrophy)
Atrophy is the shrinkage of the cells forming the glands. The cytoplasm shrivels down, leaving essentially rows of nuclei outlining the lumens (Figure 11.9). At low power these atrophic glands can look small and irregular, which may be suspicious. However, the lumens have an angular, almost staghorn look to them. Small- to medium-sized nucleoli may be seen, but the lack of cytoplasm should be a red flag against diagnosing cancer. Immunostains highlight a basal cell layer.
萎缩时,腺体中的细胞缩小。细胞质变少,几乎只留下一排核,勾勒出腺腔的轮廓(图11.9)。低倍镜下,这些萎缩的腺体小而不规则,令人怀疑恶性腺体。然而,管腔成角,看起来几乎是鹿角形。可见小到中等大小的核仁,但是缺乏细胞质,应该是不诊断癌的一个红色警戒。免疫染色显示基底细胞层存在。
基底细胞增生(Basal Cell Hyperplasia)
The basal cells that underlie the glandular cells are not usually well visualized. When they are noticeable, you can see them as sort of denim-blue, oval, regular nuclei surrounding the more purple glandular nuclei (Figure 11.10). The tricky part is that they may have nucleoli. In basal cell hyperplasia, the basal layers may proliferate and create several layers of worrisome-looking cells in the glands. The key is in recognizing the dual population; sometimes you can still see the glandular cells floating on top of the basal hyperplasia. Stains help.
腺细胞下方的基底细胞并非总能看清楚。当它们很明显时,你可以看到它们是一种牛仔布蓝色、椭圆形、规则的核,围绕在更加发紫的腺体核的周围(图11.10)。棘手的问题是它们可能有核仁。在基底细胞增生中,基底层可能会增殖,并在腺体中形成多层令人担忧的细胞。关键是识别两群细胞;有时你仍然可以看到腺体细胞漂浮在基底增生的顶部。免疫染色有帮助。
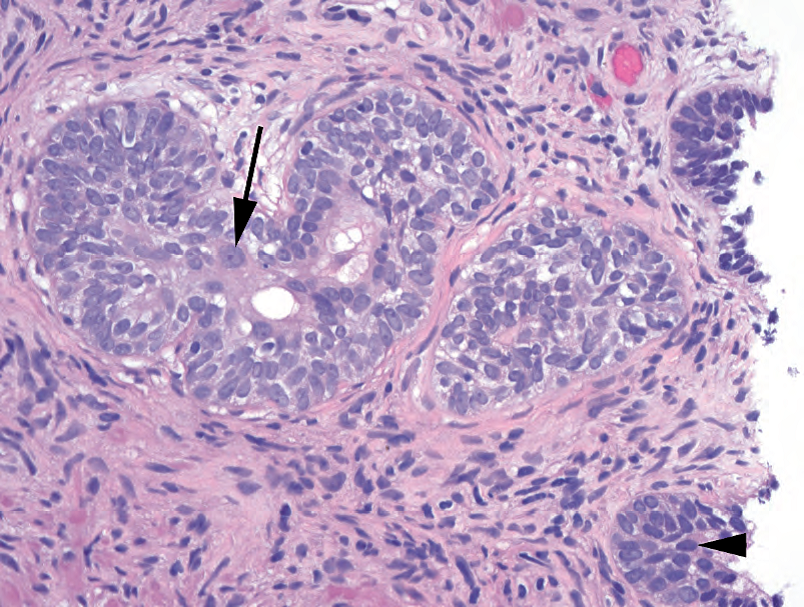
Figure 11.10. Basal cell hyperplasia. This proliferation of cells, some with prominent nucleoli (arrow), is actually an expanded basal cell layer. Comparison with benign epithelium (arrowhead) shows the relatively pale and greyish nuclei of the basal cells.
图11.10.基底细胞增生。这些增殖细胞中,部分细胞有显著核仁(箭),实际上是扩大的基底细胞层。与良性上皮(箭头)相比,基底细胞核相对淡染和灰色。
Cowper腺(Cowper’s Glands)
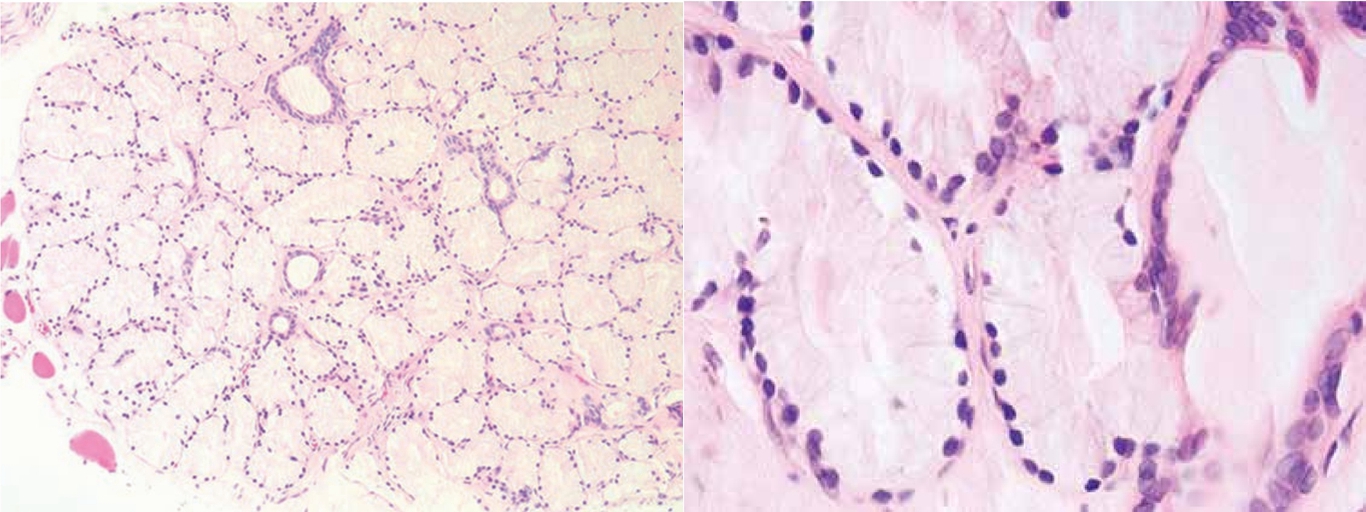
Cowper’s glands are histologically normal glands (distal to the prostate, secreting directly into the urethra, and normally not sampled on needle biopsy) that consist of mucous-filled secretory glands surrounding a coil of ducts. They are lobular in architecture and have small bland nuclei. Their abundant mucin will stain with periodic-acid Schiff, and they are usually negative for the prostate markers prostate-specific antigen (PSA) prostate-specific acid phosphatase (PSAP).
Cowper腺是组织学上正常的腺体(前列腺远端,直接分泌到尿道,粗针穿刺活检通常取不到),由黏液分泌腺和导管组成。它们呈小叶状结构,核小而温和。它们含有丰富的黏液,PAS染色阳性,而前列腺标记物(PSA和PSAP)阴性。
(译注:Cowper腺即尿道球腺,相当于女性的前庭大腺。补充图片如下:左图:低倍,穿刺活检标本可能发现横纹肌(左侧);右图:高倍。)
辐射改变(Radiation Changes)
Radiation atypia has a characteristic look that is difficult to describe in words. The nuclei are too pleomorphic to be cancer, especially when compared with the relatively uniform nuclei of prostate cancer. Radiated benign glands show atrophic cytoplasm and wildly pleomorphic nuclei mixed in with normal nuclei (Figure 11.11). The nuclei may be very large, with angular shapes, and tend to have a dense smudgy chromatin without nucleoli. Identifying residual cancer in the radiated prostate is a diagnosis made largely on architecture (many individual cells with ample vacuolated cytoplasm and nuclei that ironically are often not as pleomorphic as the benign radiated nuclei).
辐射异型性是难以描述的特征性外观。核多形性太大,超出癌的程度,尤其是与前列腺癌相对均匀的核相比。放射后良性腺体显示萎缩的细胞质和广泛的多形性核与正常核混合(图11.11)。核可能非常大,成角,并且有深染的污浊的染色质,没有核仁。放射后前列腺中的残留癌的识别,主要是根据结构进行诊断(许多单个细胞具有丰富的空泡状细胞质,而核多形性却不如良性放射性核,讽刺还是意外?)。
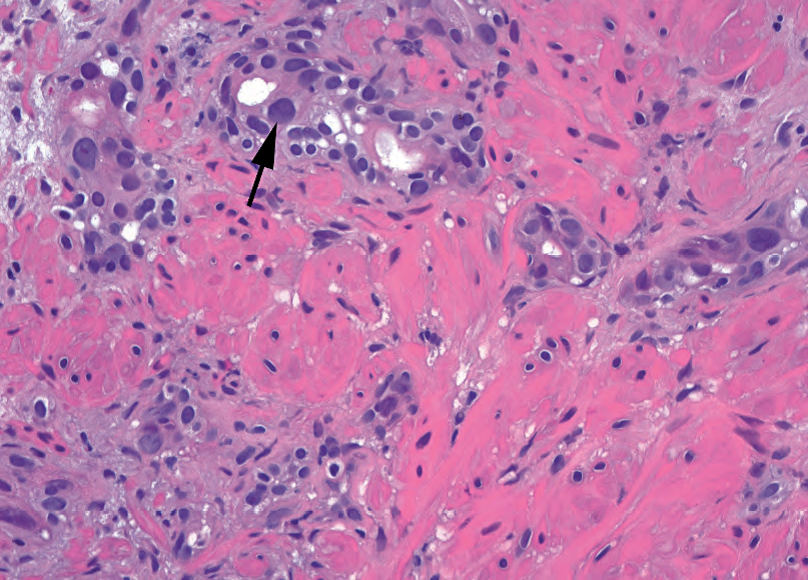
Figure 11.11. Radiation atypia in benign prostate. There is scattered and random nuclear pleomorphism (arrow). Enlarged nuclei classically have dense, uniform, smudgy chromatin.
图11.11.良性前列腺的辐射异型性。存在散在的和随机的核多形性(箭)。增大的核通常具有深染、均匀、污浊的染色质。
精囊(Seminal Vesicle)
The nuclei of the seminal vesicles have very pleomorphic nuclei, not unlike radiation atypia. They will definitely stand out in a needle biopsy specimen and can be very concerning based on cytology. However, remember that prostate cancer is usually not pleomorphic, and look for the telltale golden globs of lipofuscin to identify it as seminal vesicle (Figure 11.12).
精囊细胞具有非常明显的核多形性,与辐射异型性没有什么不同。它们肯定会在粗针穿刺活检标本中鹤立鸡群,其细胞学形态会非常令人担忧。然而,记住前列腺癌通常不是多形性的,并寻找脂褐素的金色小球来确定它是精囊(图11.12)。
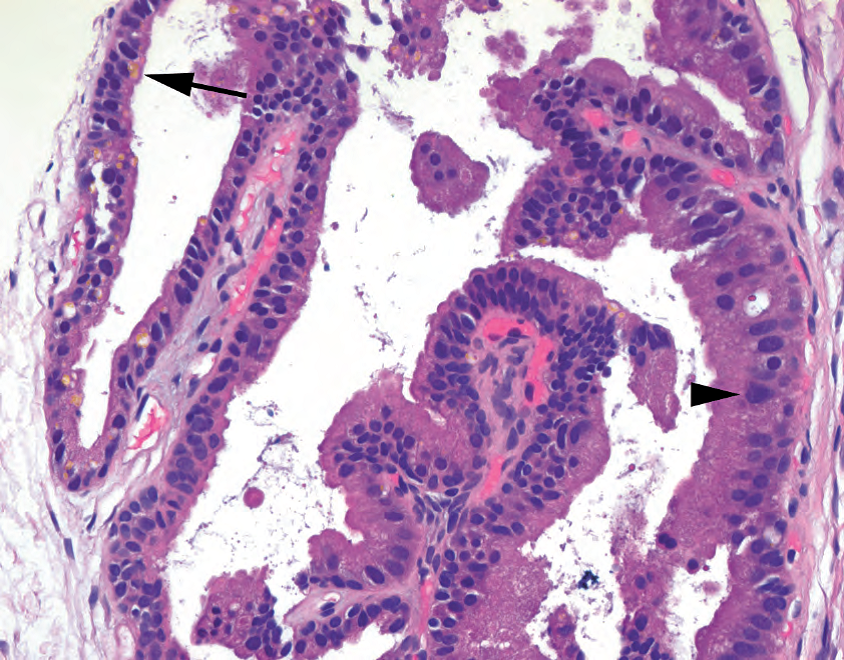
Figure 11.12. Seminal vesicle in a biopsy specimen. There are scattered large, hyperchromatic, and crowded nuclei in this gland (arrowhead). However, golden pigment is visible in the cytoplasm (arrow), identifying this as seminal vesicle.
图11.12.活检标本中的精囊。这个腺体中有散在的大的、深染的、拥挤的核(箭头)。然而,胞质中可见金黄色的色素(箭),表明这是精囊。
硬化性腺病(Sclerosing Adenosis)
Sclerosing adenosis is seen best in transurethral resection specimens. It is a hyperplastic and proliferative lesion that is complicated by a hypercellular stroma. The appearance is that of crowded glands and individual cells (which may have prominent nucleoli) in a background of cellular stroma. Remember that prostate cancer does not induce a stromal reaction.
硬化性腺病在经尿道电切标本中容易观察。它是一种增生性和增殖性病变,加上富细胞性间质。表现为富细胞性间质背景下拥挤的腺体和单个细胞(可能有显著的核仁)。记住前列腺癌不会引起间质反应。
非典型腺体和染色(Atypical Glands and Stains)
It is not uncommon, in a needle core biopsy specimen, to stumble across one or two isolated glands that make you very nervous. However, unless several features of carcinoma are evident, most pathologists will be reluctant to make the diagnosis of cancer in that setting. One option is to sign it out as a focus of atypical glands. In the absence of definitive cancer, it will usually generate a repeat biopsy.
在粗针穿刺活检标本中,偶然发现一两个孤立的腺体让你非常紧张,这种情况并不少见。然而,除非癌的几个特征是明显的,否则大多数病理医师不会在这种情况诊断癌。一种选择是报告为局灶非典型腺体。在没有明确的癌的情况下,通常会促使临床进行重复活检。
Immunostains may help in the diagnosis of these tiny lesions or with larger groups of glands that have some but not all of the features of cancer. Stains for the basal layer (CK903 or high-molecular-weight cytokeratin and p63) should highlight the basal cells in all benign glands and show loss of staining in malignant glands. Racemase is a newer marker that preferentially stains the cytoplasm of cancer. However, there are false-positive and false-negative results with all three of these antibodies, and so each case is interpreted in the context of the HE appearance (which is the best approach for all immunostains).
对于这些微小病变,或对于具有部分但非全部癌特征的较大群腺体,免疫染色可能有助于诊断。基底细胞层染色(CK903或高分子量CK和p63)应当能突出显示所有良性腺体中的基底细胞,并在恶性腺体中显示其染色缺失。消旋酶是一种新的标记物,优先染色癌细胞的细胞质。然而,这三种抗体都有假阳性和假阴性结果,因此每个病例都要结合HE表现来判读(这是所有免疫染色的最佳方法)。
(译注:CK903是一种高分子量CK,即34βE12。上文所谓“高分子量CK”实际工作中一般用CK5/6。消旋酶的全称是α-甲酰辅酶A消旋酶(AMACR,P504S)
前列腺癌根治术标本的探讨(Approach to the Radical Prostatectomy Specimen)
For the radical prostatectomy specimen, the prostate is inked and bread loafed from apex (nearest the penile urethra) to base (nearest the bladder neck). Each slice is cut into four quadrants to fit them into cassettes. The margins are taken first:
根治性前列腺切除术标本,涂抹墨汁,从顶端(最靠近阴茎尿道)到底部(最靠近膀胱颈),切成组织薄片(就像面包片)。每个组织薄片再切割成4个象限,以便装入包埋盒。取材首先取切缘:
Right and left vas deferens: These represent the true surgical margins of the vas deferens. Positive vas deferens margins are very rare, and some pathologists do not submit them.
右侧和左侧输精管:它们代表输精管的真正手术切缘。输精管切缘阳性非常罕见,有些病理医师不予报告。
Apical, or distal, margin: This is where the prostate meets the penile urethra. It is cut off as a thick tangential shave and then turned 90°, sliced, and submitted as a series of perpendicular sections that are parallel to the urethra. The presence of malignant glands is acceptable, as long as they do not touch the inked apical surface.
顶端或远端切缘:前列腺与阴茎尿道相接的地方。削取一块厚组织,然后旋转90°,切成薄片,平行于尿道、垂直连续切成组织块,依次放入包埋盒。恶性腺体的存在是可以接受的,只要它们不接触墨染的顶端表面。
Bladder neck margin: This is where the prostate meets the bladder; it is a soft tissue margin, not a urethral margin. The urethra itself retracts back into the prostate at surgery and may not be seen on the slide. This is usually a shave margin, in which the presence of any malignant glands is considered a positive margin. It can also be treated like the apical margin, however, and sectioned perpendicularly.
膀胱颈切缘:前列腺与膀胱交界处;这是一个软组织切缘,不是尿道切缘。手术时尿道本身缩回前列腺,在切片上可能看不到。通常削取切缘,任何恶性腺体的存在都被视为阳性切缘。然而,它也可以像顶端切缘一样处理,并垂直切片。
After evaluating the margins, systematically examine each section of the prostate. Each full slice of prostate is halved into left and right sides. If the entire hemisection cannot fit into one block (usually it cannot), it is subdivided into anterior and posterior quadrants. Orienting the isolated quadrant can be tricky. For posterior sections, the true posterior surface should be flatter than the lateral surface. The neurovascular bundles, which sometimes come out with the prostate, are located at the posterolateral corners. For anterior sections, the anterior tip should have many smooth muscle bundles and a very poorly defined capsule. The verumontanum of the urethra (the bump on the posterior urethra) points anteriorly (Figure 11.13).
评估切缘后,系统地检查前列腺的每个部分。每个完整的前列腺切片被分成左右两侧。如果整个半切面不能放入一个包埋盒中(通常不能),则将其细分为前后象限。确定孤立象限的方向可能很棘手。对于后切面,真实后表面应比侧面平坦。神经血管束有时与前列腺一起出现,位于后外侧角。对于前切面,前尖应该有许多平滑肌束和非常不清晰的被膜。尿道精阜(后尿道上的隆起物)指向前方(图11.13)。
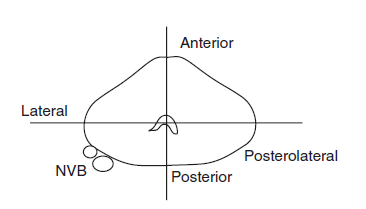
Figure 11.13. Low-power view of radical prostatectomy sections. Each cross section of prostate is cut into quadrants to fit into cassettes. The neurovascular bundles (NVB) are found at the posterolateral border of the prostate. The verumontanum points anteriorly.
图11.13.根治性前列腺切除术的低倍观,示意图。前列腺的每一个横截面都被切成4个象限,以便装入包埋盒。神经血管束(NVB)位于前列腺后外侧缘。精阜指向前方。
图中文字:
Anterior,前
Posterior,后
Lateral,外侧
Posterolateral,后外侧
Examination of the edge of the prostate is prognostically important. There is no true organ capsule but rather the outer limit or edge of the prostate, which is best recognized posteriorly and posterolaterally as packed muscle bundles of the prostate. Extension of the cancer beyond the edge indicates extraprostatic extension (EPE) and increases the stage of the tumor. Capsular incision, where the surgeon has cut across the organ and left some prostate in the patient, becomes very important if there is cancer in the area. Malignant glands near big vessels or among skeletal muscle are not necessarily EPE, but cancer in fat is “out” by definition (Figure 11.14), as there is no intraprostatic fat. However, if you wait to diagnose EPE until seeing tumor in fat, you will miss some EPE. As cancer extends beyond the border of the prostate it is often associated with a fibrotic response to the tumor, wiping out the fat. This type of EPE is better appreciated at low power by following the contour of the edge of the prostate. Anteriorly, the muscle bundles are loose and disorganized, so it is difficult to recognize EPE except by seeing tumor in or beyond the plane of adjacent fat.
前列腺切缘的检查对预后很重要。没有真正的器官被膜,而是前列腺的外部界限或边缘,从后面和后侧面来看,它是前列腺的致密肌束。癌向边界延伸并超出边界,表明前列腺外扩散(EPE),肿瘤分期升高。被膜切开是外科医生切开器官并在患者体内留下一些前列腺,如果该区域有癌,则切缘变得非常重要。大血管附近或横纹肌之间的恶性腺体不一定是EPE,但脂肪中的癌根据定义是前列腺“外”的(图11.14),因为没有前列腺内脂肪。然而,如果你等到看到脂肪中的肿瘤才诊断EPE,你会错过一些EPE。当癌扩散到前列腺边界以外时,它通常伴有肿瘤的纤维化反应,从而清除脂肪。这种类型的EPE,通过沿着前列腺边界的轮廓,在低倍下更容易识别。在此之前,肌肉束是松散和无序的,因此很难识别EPE,除非在邻近脂肪平面之内或之外看到肿瘤。
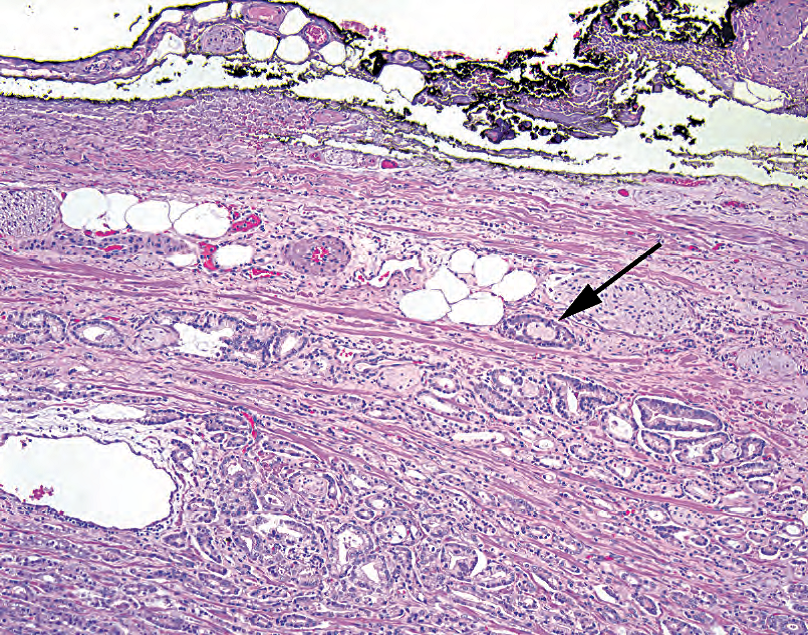
Figure 11.14. Extraprostatic extension. Malignant glands are seen wrapping around a nerve (arrow) adjacent to extraprostatic fat, diagnostic of focal extraprostatic extension. The margin, seen as the ink at the top of the photograph, is negative.
图11.14.前列腺外扩散扩散。恶性腺体包裹在靠近前列腺外脂肪的神经(箭头所示)周围,诊断为局限性前列腺外扩散。照片顶部的墨染切缘是阴性。
Although calling it “out” changes the tumor stage, it does not necessarily mean a positive margin. To call a positive margin, you must have glands not just really, really close to ink but actually transected by ink. The threshold is very high. In a positive margin, you must also decide “why” it is positive—by noting if it occurs in an area of capsular incision or in an area of EPE. A positive margin is almost always going to be considered EPE in the anterior prostate, because the capsule is so poorly defined.
尽管称之为前列腺“外”扩散会改变肿瘤的分期,但并不一定意味着肿瘤切缘呈阳性。要称之为阳性切缘,你看到的腺体不仅必须非常非常接近墨染切缘,而且必须横穿墨染切缘。诊断门槛很高。在阳性切缘中,你还必须注意它是发生在被膜切开区域或EPE区域来确定“为什么”为阳性。在前列腺前部,阳性切缘几乎总是认为EPE,因为被膜的边界很不明显。
Perineural invasion is a big deal in a biopsy specimen but is taken for granted in a radical. It is not worth mentioning in the diagnosis. Grading in a radical is the same as grading in a needle core. You only have to grade the one or two biggest nodules (not the little multifocal ones), and each big nodule is given its own grade.
神经周围侵犯在活检标本中是一个大问题,但在根治术中被认为是理所当然的。在诊断中不值得一提。根治术的分级与穿刺活检的分级相同。你只需要对一个或两个最大的结节(不是小的多灶结节)进行分级,每个大结节都有自己的分级。
The seminal vesicles are examined by sampling the seminal vesicle at the point where it meets the prostate. Seminal vesicle invasion, if present, is seen on this section. Microscopically, you need to see tumor in the parenchyma of the seminal vesicle, not just next to it.
在精囊与前列腺接触处取材检查精囊。如有精囊侵犯,可在切片中看到。显微镜下,你需要看到精囊实质中的肿瘤,而不仅仅是它旁边。
其他前列腺肿瘤(Other Prostate Neoplasms)
Ductal adenocarcinoma is a variant type of adenocarcinoma that is characterized by tall, stratified columnar cells making papillary or cribriform structures (Figure 11.15). They may grow into the urethra as exophytic masses, or they may arise from more peripheral ducts in the prostate. It may be found in conjunction with conventional adenocarcinoma. It is not assigned a Gleason grade but behaves like a pattern 4 lesion.
导管腺癌是腺癌的一种变异型,其特征是复层化高柱状细胞形成乳头状或筛状结构(图11.15)。它们可能以外生肿块的形式生长,进入尿道,也可能发生在前列腺的更外围的导管中。它可能与传统的前列腺癌同时发现。它不适用Gleason分级,但生物学表现像模式4。
Other types of carcinoma include mucinous carcinoma, squamous cell carcinoma, urothelial (transitional cell) carcinoma, sarcomatoid carcinoma, basal cell carcinoma, and small cell carcinoma. The more difficult-to-recognize variants of usual prostate cancer are pseudohyperplastic carcinoma (a sneaky variant that mimics the papillary architecture of benign hyperplasia), atrophic cancer (mimicking benign atrophy), and foamy gland cancer (with abundant xanthomatous appearing cytoplasm).
其他类型的癌包括黏液癌、鳞状细胞癌、尿路上皮(移行细胞)癌、肉瘤样癌、基底细胞癌和小细胞癌。较难识别的普通型前列腺癌的变异型包括假增生性癌(一种隐匿的变异型,假冒良性增生的乳头状结构)、萎缩性癌(假冒良性萎缩)和泡沫状腺癌(出现大量黄瘤样细胞质)。
Spindle cell lesions may arise in the prostate. Stromal lesions arising from the unique stroma of the prostate range from the benign stromal nodules, to stromal tumors of uncertain malignant potential, to stromal sarcomas. The most common prostatic sarcoma in adults is leiomyosarcoma. Rhabdomyosarcomas occur mainly in children.
前列腺可能出现梭形细胞病变。前列腺特有间质引起的间质病变范围从良性间质结节,到恶性潜能不确定的间质肿瘤,再到间质肉瘤。成人最常见的前列腺肉瘤是平滑肌肉瘤。横纹肌肉瘤主要发生在儿童。
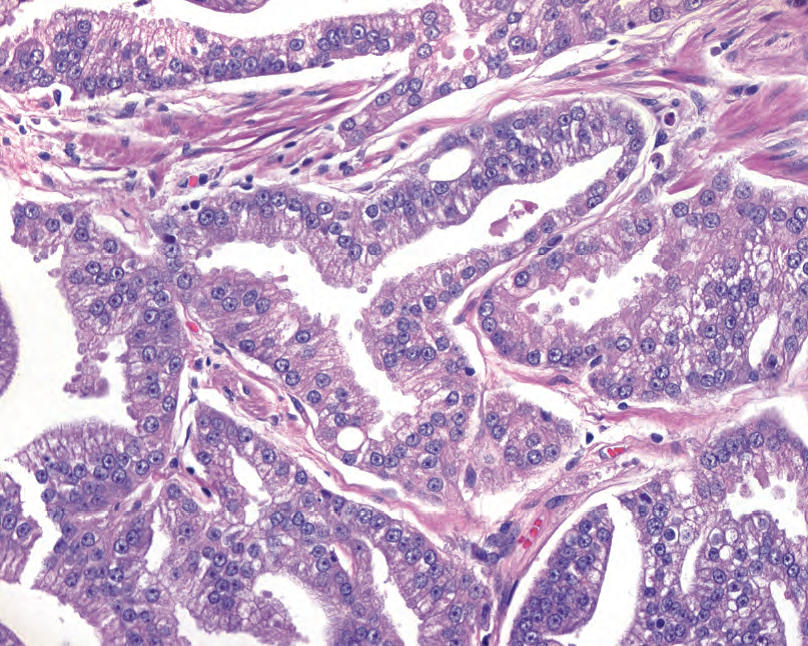
Figure 11.15. Ductal adenocarcinoma. In this variant, the tumor cells have a tall columnar morphology. The nuclei still resemble conventional prostate adenocarcinoma.
图11.15.导管腺癌。在这种变异型中,肿瘤细胞具有高柱状形态。核仍然类似于传统的前列腺癌。
来源:
The Practice of Surgical Pathology:A Beginner’s Guide to the Diagnostic Process
外科病理学实践:诊断过程的初学者指南
Diana Weedman Molavi, MD, PhD
Sinai Hospital, Baltimore, Maryland
ISBN: 978-0-387-74485-8 e-ISBN: 978-0-387-74486-5
Library of Congress Control Number: 2007932936
© 2008 Springer Science+Business Media, LLC
仅供学习交流,不得用于其他任何途径。如有侵权,请联系删除。
本站欢迎原创文章投稿,来稿一经采用稿酬从优,投稿邮箱tougao@ipathology.com.cn
相关阅读
 数据加载中
数据加载中
我要评论

热点导读
-

淋巴瘤诊断中CD30检测那些事(五)
强子 华夏病理2022-06-02 -
【以例学病】肺结节状淋巴组织增生
华夏病理 华夏病理2022-05-31 -

这不是演习-一例穿刺活检的艰难诊断路
强子 华夏病理2022-05-26 -

黏液性血性胸水一例技术处理及诊断经验分享
华夏病理 华夏病理2022-05-25 -

中老年女性,怎么突发喘气困难?低度恶性纤维/肌纤维母细胞性肉瘤一例
华夏病理 华夏病理2022-05-07







共0条评论