外科病理学实践:诊断过程的初学者指南 | 第10章 胰
第10章 胰(Pancreas)
Whipple手术(The Whipple Procedure)
The Whipple procedure is, at minimum, a pancreaticoduodenectomy, which may or may not also include pylorus of the stomach and the gallbladder. In the pylorus-preserving Whipple procedure, the simplest version, you receive the segment of duodenum from just past the pylorus to about 20 cm beyond the ampulla of Vater. The head of the pancreas is nestled in the curve of the duodenum near the ampulla; the pancreas is shaped like a J, and the head is the base of the J, with the uncinate process as the hook. The distal common bile duct runs through the pancreas and enters the ampulla, where it is joined by the main pancreatic duct (Figure 10.1). Usually it is only the head of pancreas that comes out; if the tail is also involved, you may get the total pancreas and spleen.
Whipple手术至少是胰十二指肠切除术,可能包括或不包括胃幽门和胆囊。在保留幽门的Whipple手术中,最简单的术式是从刚过幽门到Vater壶腹外约20厘米的十二指肠段。胰头位于壶腹附近的十二指肠弯曲处;胰腺呈J形,胰头为J形的基部,钩突为弯钩处。远端胆总管穿过胰腺进入壶腹,在壶腹与主胰管相连(图10.1)。通常只有胰头露在外面;如果胰尾也受累,你可能会得到整个胰腺和脾。
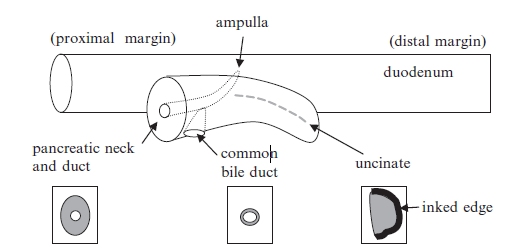
Figure 10.1. Diagram of specimen obtain during a Whipple procedure. The head of the pancreas comes out attached to a segment of duodenum. The main pancreatic duct is visible at the pancreatic neck margin (a surgical margin). The common bile duct enters the pancreas to join the pancreatic duct (also a surgical margin). The uncinate process is the tip of the pancreas, and its edge abuts major vessels (a soft tissue margin).
图10.1.Whipple手术标本示意图。露在外面的胰头与一段十二指肠相连。主胰管在胰颈切缘(手术切缘)可见。胆总管(也是一个外科切缘)进入胰腺与胰管相连。钩突在胰腺的一端,其切缘邻接大血管(软组织切缘)。
图中文字:
proximal margin,近端切缘
ampulla,壶腹
distal margin,远端切缘
duodenum,十二指肠
pancreatic neck and duct,胰颈和胰管
common bile duct,胆总管
uncinate,钩突
inked edge,墨染切缘
There are five principal margins that are usually sampled on frozen section (see Figure 10.1). The first is the pancreatic margin, or the pancreatic neck (where the J is transected). This is usually taken as a shave margin, sampling the entire cross section of pancreas, and cancer anywhere on the slide is a positive margin. There is no neck margin on a total pancreatectomy.
冰冻切片通常要取5个主要切缘(见图10.1)。第一个是胰腺切缘或胰颈切缘(J形被切断的地方)。这里通常削取切缘,取胰腺的整个横截面,切片上任何位置有癌都视为阳性切缘。全胰腺切除术没有胰颈切缘。
The second margin is the common bile duct margin, which is a shave of the bile duct stump. This ensures that cancer is not tracking up the bile duct toward the liver.
第二个切缘是胆总管切缘,这是胆管残端,也是幻灯片。这确保癌不会沿着胆管向肝转移。
The third is the uncinate margin. This is the tip of the short end of the J, and it represents the place where the pancreas sits against the major vessels. For the uncinate, you should take one representative perpendicular margin, and the edge of the tissue is inked. Cancer on the slide is okay, as long as it does not touch ink. As this tissue abuts major vessels, the surgeon often cannot resect additional tissue anyway.
第三个是钩突切缘。这是J形短端的尖端,代表胰腺靠着大血管的位置。对于钩突,切取一个有代表性的垂直切缘,并且组织切缘已涂抹墨水。切片上有癌是可以的,只要它不接触墨水。由于该组织毗邻大血管,外科医生通常无法切除更多的其他组织。
The fourth and fifth margins are the proximal and distal duodenal margins. It is rare for these sections to contain tumor.
第四和第五个切缘是十二指肠近端和远端切缘。这些切片很少含有肿瘤。
Most Whipple procedures are performed for a pancreatic mass seen radiologically. Although it is possible to get a cancer diagnosis by fine-needle aspiration, this is not always performed, and false-negative results are not uncommon. Therefore, often our first look at the tumor is during the Whipple procedure.
大多数Whipple手术是针对放射学看到的胰腺肿块。虽然可以通过细针穿刺获得癌诊断,但并不总是这样,假阴性结果并不少见。因此,我们通常在Whipple手术标本第一次看到肿瘤。
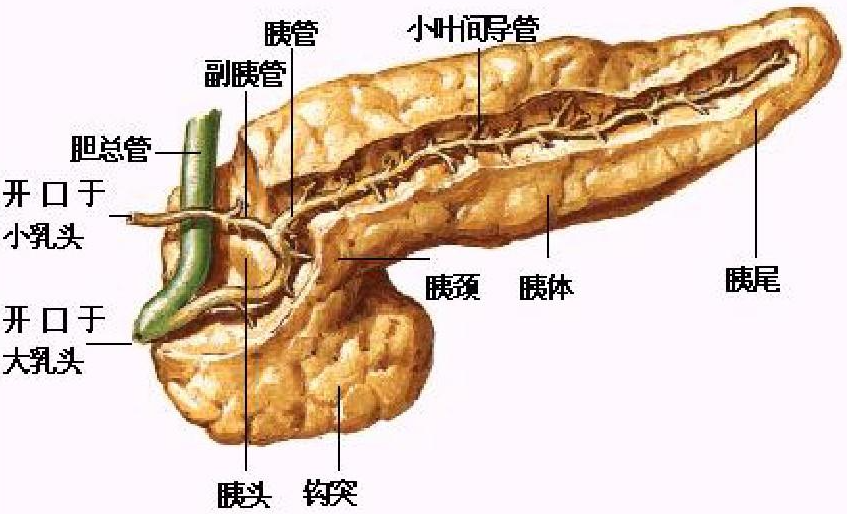
(译注:补充胰腺解剖示意图如下)
 正常胰腺(The Normal Pancreas)
正常胰腺(The Normal Pancreas)
The normal pancreas is a large mixed exocrine and endocrine gland, with acinar cells arranged around ducts in lobular units. The acinar cells secrete digestive enzymes in precursor forms, which travel to the duodenum via the ducts. Normal ducts are low cuboidal epithelium, and the acinar cells are wedge-shaped granular pink and purple cells (Figure 10.2). Scattered among them are the neuroendocrine islets of Langerhans, which show typical neuroendocrine cytology and are arranged in little nests.
正常胰腺是一个巨大的外分泌和内分泌混合腺,腺泡细胞排列在导管周围,形成小叶单位。腺泡细胞分泌消化酶前体,通过导管进入十二指肠。正常导管为低立方上皮,腺泡细胞为楔形颗粒性粉红色和紫色细胞(图10.2)。其中散布着Langerhans神经内分泌小岛(译注:即胰岛),显示出典型的神经内分泌细胞学,排列成小巢状。
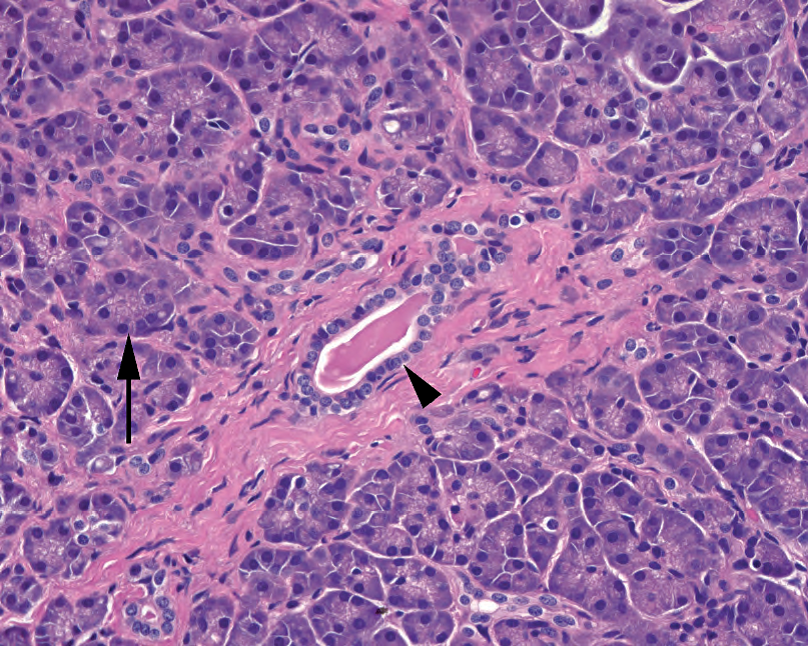
Figure 10.2. Normal pancreatic acinus. The duct is seen at the center (arrowhead), with surrounding acini of secretory cells (arrow).
图10.2.正常胰腺腺泡。导管位于中心(箭头),周围有分泌细胞的腺泡(箭号)。
慢性胰腺炎与导管腺癌(Chronic Pancreatitis Versus Ductal Adenocarcinoma)
Chronic pancreatitis is not an uncommon finding in a resected pancreas. The damage done to the pancreas by chronic obstruction, as with a mass, causes diffuse fibrosis, atrophy of the acinar units, reactive changes, and disruption of the normal architecture, all of which can mimic carcinoma. One of the hardest tasks (especially on frozen section) is differentiating reactive pancreatic ducts from well-differentiated infiltrating adenocarcinoma, the most common pancreatic malignancy. Some tips include the following:
慢性胰腺炎在切除的胰腺中并不少见。慢性阻塞(如肿块)对胰腺造成的损害,会导致弥漫性纤维化、腺泡单位萎缩、反应性改变和正常结构破坏,所有这些都可能貌似癌。最困难的任务之一(尤其是冰冻切片)是区分反应性胰腺导管和高分化浸润性腺癌,后者是最常见的胰腺恶性肿瘤。一些提示包括:
有用但主观(Helpful but subjective)
On low power, chronic pancreatitis has a lobular architecture, with large central ducts surrounded by smaller peripheral ones. Cancer is haphazard, with random and irregular distribution of glands (Figure 10.3).
低倍镜下,慢性胰腺炎具有小叶结构,大的中央导管围绕着小的外周导管。癌是杂乱的,腺体随机地不规则分布(图10.3)。
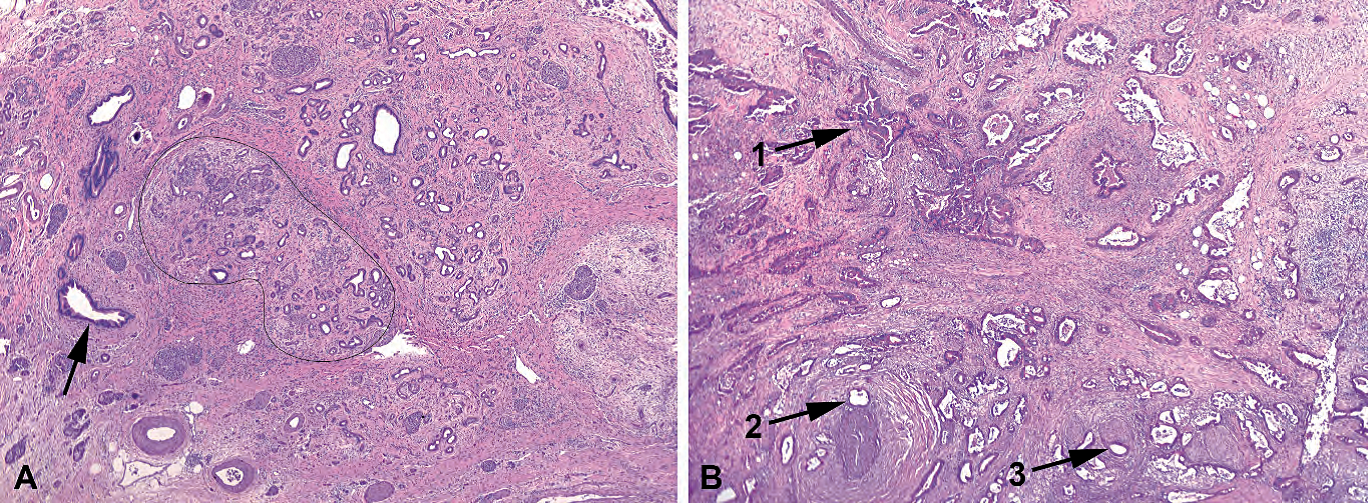
Figure 10.3. Chronic pancreatitis versus cancer, low power. (A) In chronic pancreatitis, the large ducts may show marked reactive changes, appearing blue and prominent, but they should still be located between lobules of acini (arrow). The acini show marked atrophy and fibrosis such that only the small ducts remain and appear infiltrative; however, the lobules retain a circumscribed outline (black line). (B) In adenocarcinoma, large, prominent, irregularly shaped ducts are scattered throughout, without respect to normal architecture (1). Large ducts next to vessels (2) or nerves (3) are diagnostic of cancer.
图10.3.慢性胰腺炎与癌,低倍。(A)在慢性胰腺炎中,大导管可能显示明显的反应性改变,呈蓝色且明显,但仍应位于腺泡小叶之间(箭号)。腺泡显示明显萎缩和纤维化,只有小导管残留,像是浸润性;然而,小叶保留一个边界清楚的轮廓(黑线)。(B)在腺癌中,大的、显著的、不规则形状的导管散在遍布,与正常结构无关(1)。靠近血管(2)或神经(3)的大导管对癌有诊断意义。
Incomplete lumina, in which the luminal spaces are not symmetrically surrounded by nuclei, and luminal necrosis both point to a diagnosis of pancreatic cancer (Figure 10.4).
腺腔不完整,腺腔周围的细胞核不对称地分布;以及腺腔坏死,二者对癌均有诊断意义(图10.4)。
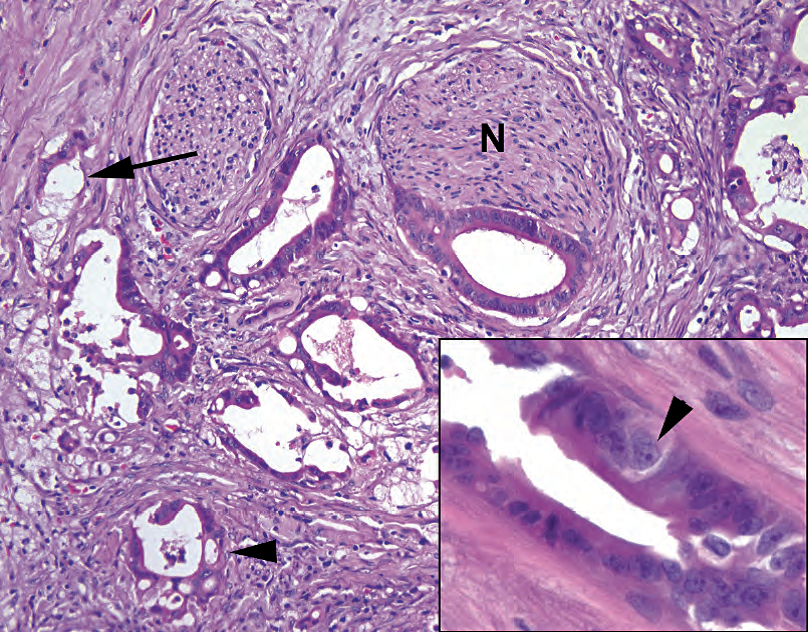
Figure 10.4. Adenocarcinoma. On high power, the infiltrative glands show incomplete lumens (arrow), cribriform growth pattern (arrowhead), and perineural invasion (N). Inset: Marked variation in nuclear size is diagnostic of cancer. Note the large nucleus with prominent nucleolus (arrowhead) across the gland from nuclei less than one fourth of its size.
图10.4.腺癌。高倍镜下,浸润腺体显示腺腔不完整(箭号)、筛状生长模式(箭头)和神经周围浸润(N)。插图:细胞核大小的显著变化对癌有诊断意义。注意大核有显著核仁(箭头),大核穿过腺体中较小的核,小核不到大核的1/4。
Note cellular pleomorphism. In general, cancer tends to have hyperchromatic and irregularly shaped nuclei, mitoses, and necrosis (see Figure 10.4). You will hear of the “4:1 rule,” which states that if, in one gland, one nucleus is four times the size (area) of another, it is cancer. However, chronic pancreatitis can lead to some more subtle atypia, and it does take some experience to tell the difference between a 4:1 ratio and a 3:1 ratio. Also, you will sometimes see well-differentiated pancreatic carcinoma with uniform nuclei.
注意细胞多形性。一般来说,癌往往有核深染和核不规则、核分裂和坏死(见图10.4)。你会听到“4:1规则”,即,如果一个腺体中的一个核的大小(面积)是另一个核的四倍,那就是癌。然而,慢性胰腺炎可能会导致某些更微妙的非典型性,要区分4:1和3:1的比例确实需要一些经验。此外,你有时会看到分化良好的胰腺癌,细胞核均匀。
无用(Not helpful)
The fibrosis of chronic pancreatitis can look much like a desmoplastic stromal response. However, the pale edematous fibrosis can accentuate the lobular architecture of chronic pancreatitis, which is helpful.
慢性胰腺炎的纤维化可能貌似促结缔组织增生性间质反应。然而,淡染水肿性纤维化可能使得慢性胰腺炎的小叶结构更明显,有助于区分。
Every intern dots all the benign islets of Langerhans on a pancreatic neck, usually missing the sneaky invasive stuff. Islets, in chronic pancreatitis, are essentially all that remains of the withered parenchyma, and therefore they look crowded, infiltrative, and haphazard (Figure 10.5). As is true for any endocrine cell, these cells can have some pleomorphism, and in some cases they can involve perineural spaces. Fortunately, the chromatin still looks neuroendocrine, so try to ignore them even though they really do look a little like lobular breast carcinoma.
每个实习生都会在胰颈画出所有良性胰岛,却通常会漏掉那些鬼鬼祟祟的浸润性东西。在慢性胰腺炎中,胰岛基本上是萎缩的胰腺实质的全部残余,因此它们看起来拥挤、浸润且杂乱分布(图10.5)。正如任何内分泌细胞一样,这些细胞可能具有某些多形性,在某些病例中,它们可能累及神经周围间隙。幸运的是,染色质看起来仍然是神经内分泌的,所以尽量忽略它们,即使它们看起来确实有点像乳腺小叶癌。
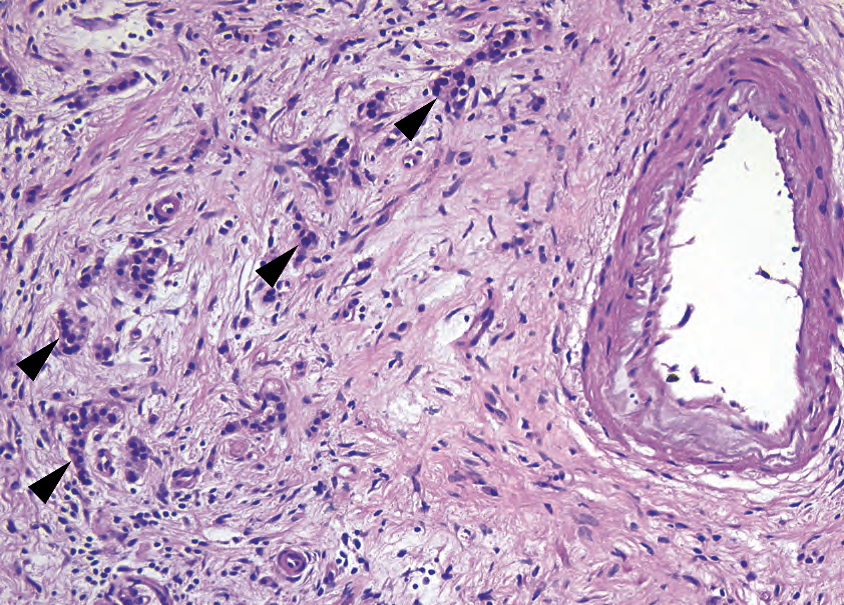
Figure 10.5. Residual islets of Langerhans. Neuroendocrine cells are among the last to go in chronic pancreatitis and appear to infiltrate through the fibrotic stroma (arrowheads). However, their small, round, dense, regular nuclei do not resemble pancreatic adenocarcinoma.
图10.5.残留的胰岛。神经内分泌细胞是慢性胰腺炎最后剩余的细胞之一,似乎通过纤维化间质浸润(箭头)。然而,它们的核小、圆、致密、规则分布,与胰腺癌不同。
免费赠品(即使是初学者也能解释)(Freebies (even the beginner can interpret them))
Glands in a nerve, or perineural invasion, always indicate cancer.
神经中的腺体,或神经周围浸润,总是提示癌。
Large ducts running next to a large muscular vessel almost always indicate cancer (Figure 10.6).
靠近肌性大血管的大导管,几乎总是提示癌(图10.6)。
Ducts leaving the pancreas to infiltrate the duodenum always indicate cancer.
离开胰腺的导管浸润十二指肠,总是提示癌。
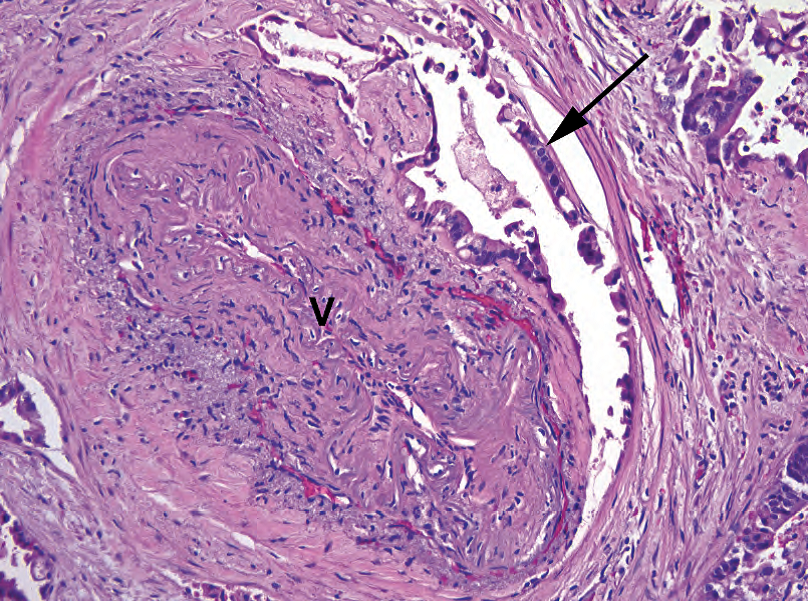
Figure 10.6. Adenocarcinoma next to a vessel. Large duct-like structures (arrow) next to a large-caliber vessel (V) are almost certainly cancer, even if deceptively well differentiated.
图10.6.血管旁边的腺癌。大口径血管(V)旁边的大导管状结构(箭号)几乎肯定是癌,即使貌似温良。
胰腺中的异型增生(Dysplasia in the Pancreas)
The pancreas is not an organ that can be evaluated with serial biopsies, and thus the natural history and malignant potential of dysplasia are not well understood. However, there are recognized grades of dysplasia within the duct system, called pancreatic intraepithelial neoplasia (PanIN). This ranges from PanIN 1, which may overlap with hyperplastic or reactive changes, to PanIN 3, which is carcinoma in situ. A lesion should always be graded by the highest level of dysplasia seen.
胰腺不能通过连续活检来评估,因此异型增生的自然史和恶性潜能还不清楚。然而,导管系统内存在公认的异型增生分级,称为胰腺上皮内肿瘤(PanIN)。其范围从可能与增生性或反应性改变重叠的PanIN 1到PanIN 3(原位癌)。始终按所见的最高异型增生程度进行分级。
PanIN 1A has a flat layer of tall columnar cells with basal nuclei and apical mucin and no atypia. The cells are similar to normal endocervical glands (Figure 10.7). PanIN 1B is the same as PanIN 1A but with a papillary or undulating appearance.
Panin 1A有一层平坦的高柱状细胞,有基底核和顶端黏液,无非典型性。这些细胞类似正常的宫颈腺(图10.7)。PanIN 1B与PanIN 1A相同,但有乳头状或起伏的结构。
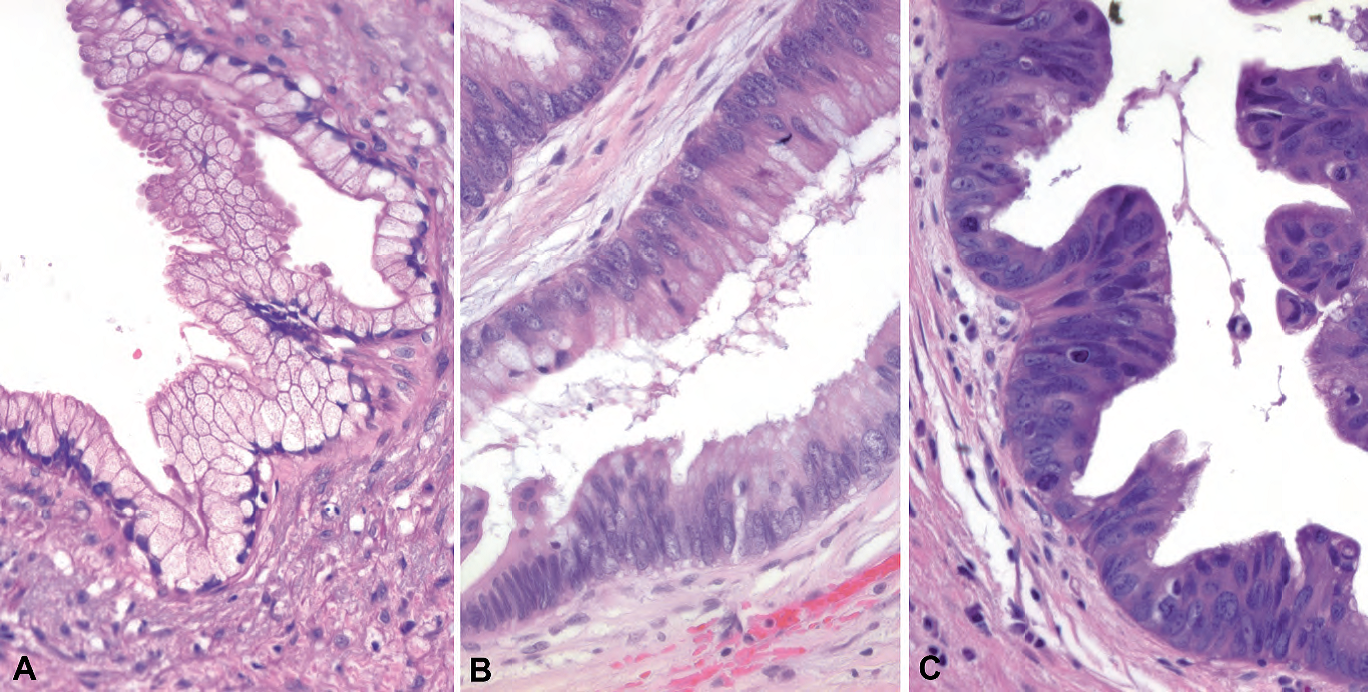
Figure 10.7. The grades of pancreatic intraepithelial neoplasia (PanIN). (A) PanIN 1 shows tall mucinous cells resembling endocervix. (B) PanIN 2 shows increasing nuclear crowding, enlargement, and atypia, suggestive of a tubular adenoma of colon. (C) PanIN 3 shows highgrade nuclei with loss of polarity, frequent mitoses, and loss of mucinous differentiation.
图10.7.胰腺上皮内肿瘤(PanIN)的分级。(A)PanIN 1显示类似子宫颈内膜的高柱状黏液细胞。(B)PanIN 2显示核拥挤、核增大和异型性增加,类似结肠管状腺瘤。(C)PanIN 3显示高级别细胞核,极性丧失,核分裂频繁,黏液分化丧失。
PanIN 2 is flat or papillary but with nuclear abnormalities, including nuclear crowding and enlargement, stratification, hyperchromasia, and sometimes basal mitoses. This epithelium should remind you of a tubular adenoma or what would be called low-grade dysplasia in the gastrointestinal tract (see Figure 10.7).
PanIN 2呈平坦或乳头状,但有核异常,包括核拥挤和增大、复层化、深染,有时有基底部核分裂。这种上皮让你想起管状腺瘤或胃肠道的低度异型增生(见图10.7)。
PanIN 3 is carcinoma in situ. You may see a cribiforming, papillary, or micropapillary architecture or necrosis. Cytologic features include large ugly nuclei with prominent nucleoli, total loss of polarity, atypical mitoses, maloriented goblet cells (upside down)—essentially the same criteria you would use for high-grade dysplasia in other gastrointestinal epithelia (see Figure 10.7).
PANIN 3是原位癌。可能会看到筛状、乳头状或微乳头状结构或坏死。细胞学特征包括丑陋的大核,核仁显著,极性完全丧失,非典型核分裂象,方向错乱的杯状细胞(倒置)—基本上与其他胃肠道上皮高度异型增生的标准相同(见图10.7)。
Invasive carcinoma arising out of PanIN 3 is well documented. However, remember that PanIN is a common incidental finding in a pancreas. It is not visible radiologically, it does not make a mass, and it does not cause obstruction. If you have a clinical mass, you should be thinking instead of an invasive carcinoma or intraductal papillary mucinous neoplasm (IPMN; see next section). Also, do not worry too much about the PanINs. With the exception of PanIN 3, they are of no proven clinical significance; margins with PanIN 1 or PanIN 2 lesions can safely be called negative.
起源于PanIN 3的浸润性癌已有大量的文献报道。然而,请记住,PanIN是胰腺中常见的偶然发现。放射学上看不见,不形成肿块,也不造成阻塞。如果有临床肿块,应该考虑的浸润性癌或导管内乳头状黏液性肿瘤(IPMN;见下一节)。另外,不要太担心PanIN。除PanIN 3外,它们没有已证实的临床意义;PanIN 1或PanIN 2病变的切缘可以安全地称为阴性。
导管内乳头状黏液性肿瘤(Intraductal Papillary Mucinous Neoplasm)
An IPMN is defined as a mucin-producing neoplasm arising in either the main pancreatic duct or a secondary (side-branch) duct. The ducts are usually dilated because they are full of a papillary proliferation and abundant mucin. The main lesion to consider in the differential diagnosis is the mucinous cystic neoplasm (discussed later). If you have a mucin-producing cystic neoplasm in the pancreas, always probe the main duct to see if the cysts are connected to it (an IPMN) or not (a mucinous cystic neoplasm). Essentially it is a gross diagnosis and may even be an endoscopic one; if mucin was seen coming out of the ampulla, the cysts must be connected to the pancreatic ducts and the lesion is more likely to be an IPMN. However, once you have identified an IPMN grossly, you must look microscopically to evaluate the level of atypia and rule out an invasive carcinoma. Intraductal papillary mucinous neoplasms are divided into three categories:
IPMN的定义是一种产生黏液的肿瘤,发生在主胰管或副胰管(侧支)。导管通常扩张,因为充满乳头状增殖和丰富的黏液。主要鉴别诊断是黏液性囊性肿瘤(稍后讨论)。如果在胰腺发现分泌黏液的囊性肿瘤,一定要探测主导管,看看囊肿与之相连(IPMN),或不相连(黏液性囊性肿瘤)。本质上,这是一个大体诊断,甚至可能是一个内窥镜诊断;如果看到壶腹流出黏液,囊肿必然与胰管相连,病变更可能是IPMN。然而,一旦你大体上确定了IPMN,你必须在显微镜下观察以评估非典型性水平并排除浸润性癌。导管内乳头状黏液性肿瘤分为三类:
With low-grade dysplasia: These neoplasms are cytologically bland and have the same criteria as PanIN 1.
伴低度异型增生:这些肿瘤的细胞形态学温和,其标准与PanIN 1相同。
With moderate dysplasia (formerly known as borderline): These neoplasms cytologically show increasing nuclear abnormalities and have the same criteria as PanIN 2.
中度异型增生(以前称为交界性):这些肿瘤细胞学显示核异常增加,其标准与PanIN 2相同。
With high-grade dysplasia: These neoplasms are cytologically malignant (see PanIN 3 criteria, discussed earlier). Any IPMN with high-grade dysplasia must be carefully scrutinized for invasive carcinoma slipping out of the duct.
伴高度异型增生:这些肿瘤细胞学呈恶性(参见前面讨论的PanIN 3标准)。任何伴有高度异型增生的IPMN都必须仔细检查是否有浸润性癌超出导管范围。
A common question is, how can I tell PanIN in a largish duct from IPMN in a smallish sidebranch duct? Features that favor an IPMN include the following:
一个常见的问题是,如何区分大导管中的PanIN和较小侧支管管道中的IPMN?倾向IPMN的特点包括:
Long papillae, or finger-like projections with fibrovascular cores (Figure 10.8)
长乳头,或指状突起,有纤维血管轴心(图10.8)
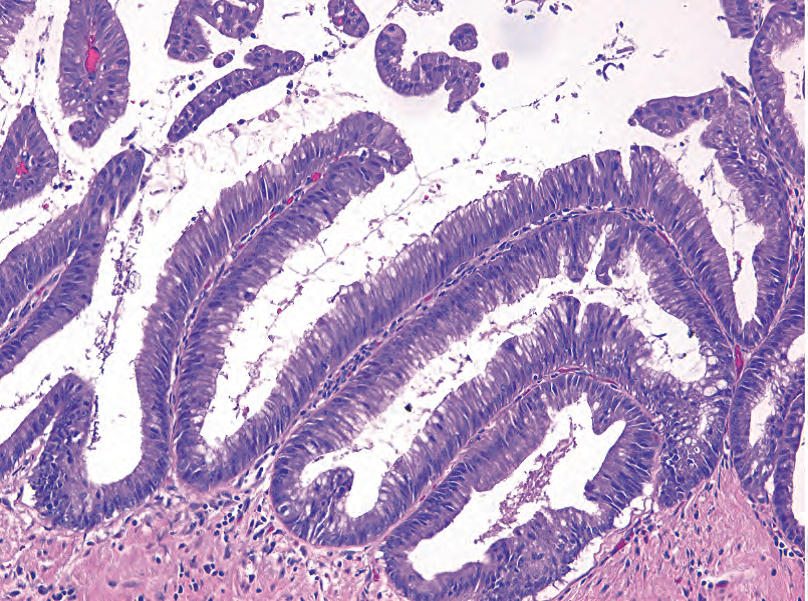
Figure 10.8. Papillary projections, intraductal papillary mucinous neoplasm with moderate dysplasia. These tall papillary fronds are covered with mucinous cells showing moderate dysplasia, similar to PanIN 2.
图10.8.乳头状突起,导管内乳头状黏液性肿瘤伴中度异型增生。这些高高的乳头结构被覆黏液细胞,显示中度异型增生,类似于PanIN 2。
Blue mucin in the lumen of the duct
导管腔中的蓝色黏液
Continuity with one of the main pancreatic ducts
与其中一条主胰管相连
Grossly or radiologically visible
肉眼可见或放射学上可见
When it comes right down to it, identifying the grade of dysplasia correctly right is much more important than distinguishing between an IPMN and a PanIN.
归根结底,正确识别异型增生的等级比区分IPMN和PanIN更重要。
浸润性导管腺癌(Invasive Adenocarcinoma (Ductal))
The most common form of infiltrating adenocarcinoma in the pancreas is ductal. It usually arises in the head and often invades adjacent structures before coming to clinical attention. The histologic features of ductal adenocarcinoma have been described earlier. Once you have established carcinoma, look carefully at all sections of duodenum and extrapancreatic bile duct to see if the carcinoma invades those structures (in increase in stage). The bile duct and ampullary region have numerous benign glands branching off of them, but remember that the benign glands will have a lobular and symmetric look at low power. Variants of ductal adenocarcinoma include adenosquamous, colloid (mucinous), hepatoid, medullary, signet ring cell, undifferentiated (anaplastic), and undifferentiated carcinoma with osteoclast-like giant cells.
胰腺中最常见的浸润性腺癌是导管腺癌。它通常发生在胰头,并在引起临床注意之前侵入邻近结构。导管腺癌的组织学特征已在前面描述过。一旦确定了癌,仔细观察十二指肠和胰外胆管的所有部分,看看癌是否侵入这些结构(分期升级)。胆管和壶腹区有许多良性腺体从中分支出来,但要记住,良性腺体在低倍镜下呈小叶状和对称外观。导管腺癌的变异型包括腺鳞癌、胶样癌(黏液癌)、肝样癌、髓样癌、印戒细胞癌、未分化癌(间变性癌),和具有破骨细胞样巨细胞的未分化癌。
胰腺其他囊性病变(Other Cystic Lesions of the Pancreas)
黏液囊性肿瘤(Mucinous Cystic Neoplasm)
The mucinous cystic neoplasm occurs almost always in middle-aged women, usually in the tail of the pancreas. This mucinous neoplasm produces multilocular cysts that do not communicate with the main duct system. They have, by definition, a rim of ovarian stroma (Figure 10.9), so think of them as mucinous ovarian tumors heterotopic into the pancreas. As in the ovary, they have three grades, and these grades conveniently parallel the three grades of the IPMN:
黏液囊性肿瘤几乎总是发生在中年女性,通常发生在胰尾。该肿瘤产生多房囊肿,与主导管系统不相连。根据定义,它们有一圈卵巢型间质(图10.9),因此可以视为异位进入胰腺的黏液性卵巢肿瘤。与卵巢一样分三级,与IPMN的三个等级平行:
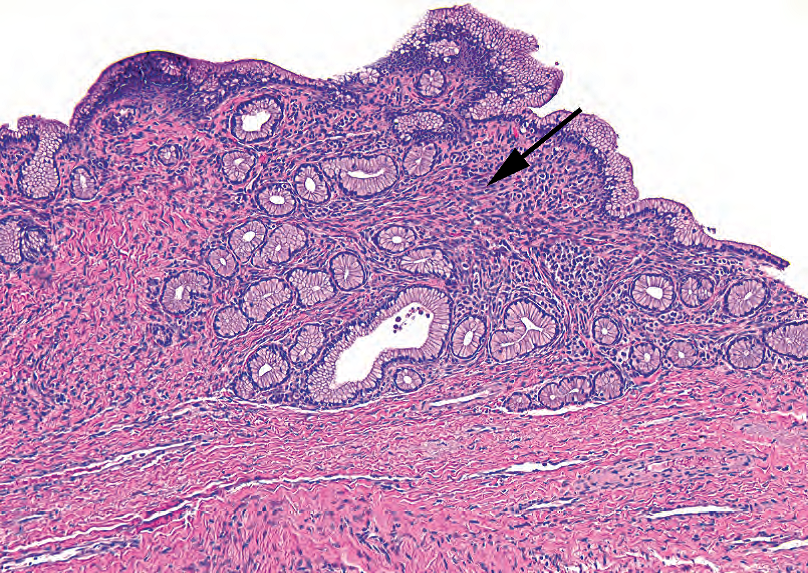
Figure 10.9. Mucinous cystic neoplasm. The cyst lining is composed of mucinous cells, benign in this example, and underlying blue spindly ovarian-type stroma (arrow).
图10.9.黏液囊性肿瘤。囊肿被覆黏液细胞,本例为良性,下面为蓝色梭形卵巢型间质(箭号)。
With low-grade dysplasia: no atypia, like PanIN 1
低伴度异型增生:无非典型性,如PanIN 1
With moderate dysplasia: increasing nuclear atypia and/or architectural complexity, like PanIN 2
伴中度异型增生:核异型性和/或结构复杂性增加,如PanIN 2
With high-grade dysplasia: carcinoma in situ, like PanIN 3
伴高度异型增生:原位癌,如PanIN 3
Approximately one third of mucinous cystic neoplasms have an associated invasive carcinoma, which would be called infiltrating moderately differentiated adenocarcinoma arising in association with a mucinous neoplasm with high-grade dysplasia.
大约1/3黏液囊性肿瘤伴有浸润性癌,称为浸润性中分化腺癌起源于并发的黏液性肿瘤伴高度异型增生。
浆液性囊腺瘤(Serous Cystadenoma)
Serous cystadenomas of the pancreas, unlike the serous cystadenomas of the ovary, are almost always microcystic. Grossly, they have a central scar and radiating small clear-fluid-filled cysts, like the cross section of a lime. Microscopically, the cysts are lined by cuboidal cells with clear cytoplasm (glycogen) and small, uniform, round nuclei (Figure 10.10). Areas of more solid or trabecular growth may look much like metastatic renal cell carcinoma, which is in fact in the differential. Serous cystadenocarcinomas exist but are extremely rare.
与卵巢浆液性囊腺瘤不同,胰腺浆液性囊腺瘤几乎都是微囊。大体上,有一个中心瘢痕和放射状分布的充满透明液体的小囊肿,像酸橙的横截面。显微镜下,囊肿被覆立方形细胞,胞质透明(糖原),核小、圆而均匀(图10.10)。更加实性或小梁生长的区域可能貌似转移性肾细胞癌,确实需要鉴别。也存在浆液性囊腺癌,但极为罕见。
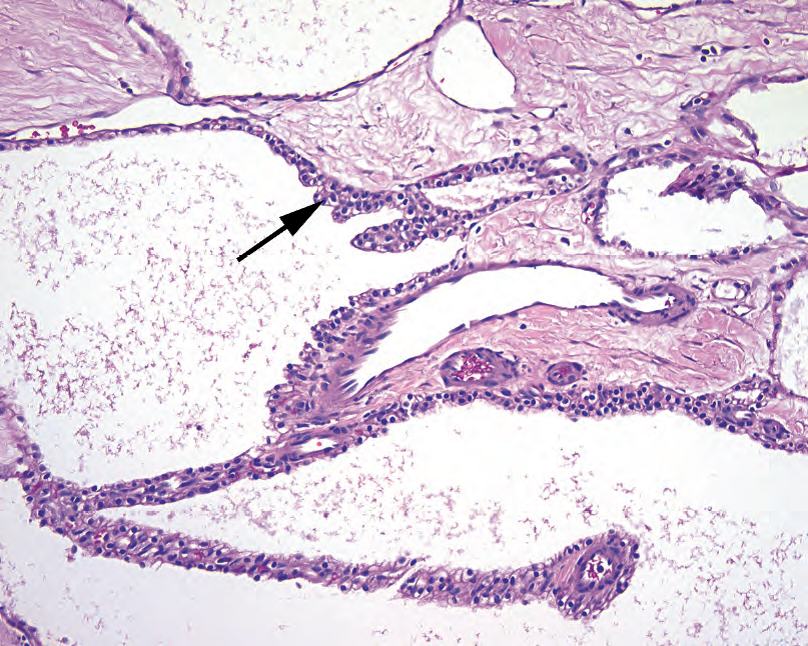
Figure 10.10. Serous cystadenoma, high power. The cells lining the multilocular cyst are small, with dense round nuclei and clear cytoplasm (arrow).
图10.10.浆液性囊腺瘤,高倍。多房囊肿的被覆细胞小,有致密的圆核和透明胞质(箭头)。
实性假乳头状肿瘤(Solid-Pseudopapillary Neoplasm)
Solid-pseudopapillary neoplasms are unusual and distinctive tumors in the differential diagnosis of cystic lesions in young women. They are malignant but extremely indolent. The cell of origin is not known, and so the neoplasm is named based on its appearance. These neoplasms start out solid but undergo cystic degeneration and therefore may present as a cyst (despite the name). The cells are characteristically noncohesive, and so the remaining solid areas show a pseudopapillary growth pattern (meaning there is solid growth along fibrovascular septa, with a dropout of the loosely cohesive cells in between septa and a resulting papillary look). The nuclei are small, oval, bland, and grooved (Figure 10.11).
在年轻女性囊性病变的鉴别诊断中,实性假乳头状肿瘤是一种少见的独特肿瘤。它们是恶性的,但极其惰性。起源细胞未知,因此根据其形态学命名。这些肿瘤开始是实性,但会发生囊性变,因此可能表现为囊性(尽管名称不同)。细胞的特征是无黏附性,因此其余的实性区域显示假乳头状生长模式(意思是沿着纤维血管间隔呈实性生长,间隔之间的细胞呈松散黏附并脱落,因此看似假乳头结构)。细胞核小,椭圆形,形态温和,有核沟(图10.11)。
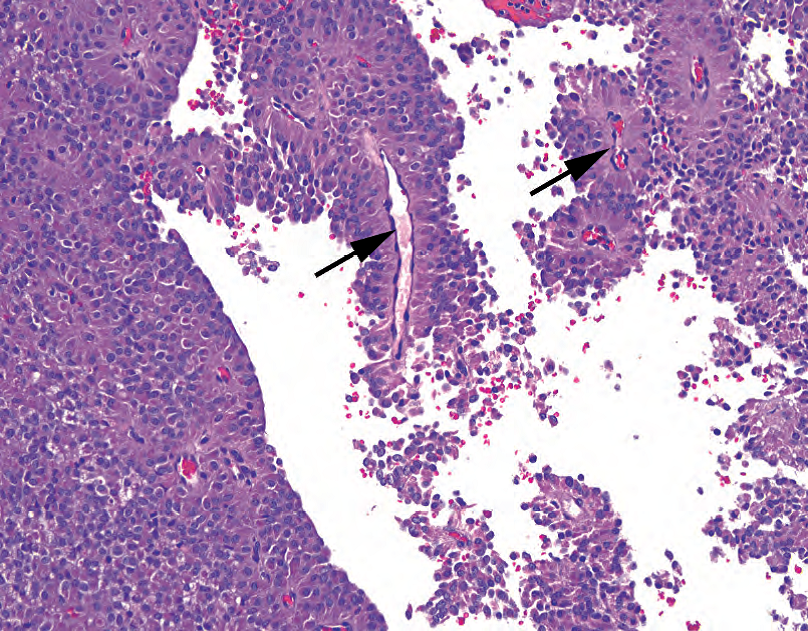
Figure 10.11. Solid pseudopapillary tumor. The small plasmacytoid cells with neuroendocrine-type chromatin could be mistaken for islet cell tumor or acinar cell carcinoma. However, this growth pattern, with rosette-like growth around fibrovascular cores (arrows) and dropout of the intervening cells, is typical of the solid pseudopapillary tumor.
图10.11.实性假乳头状肿瘤。浆细胞样小细胞伴神经内分泌型染色质,可能误诊为胰岛细胞瘤或腺泡细胞癌。然而,这种生长模式(围绕纤维血管轴心(箭号)的菊形团样生长,介于其间的细胞脱落)是实性假乳头状肿瘤的典型表现。
The differential diagnosis for this neoplasm includes well-differentiated pancreatic endocrine neoplasm and acinar cell carcinoma, both of which are discussed later. Immunohistochemical labeling is very helpful, as solid-pseudopapillary neoplasms are CD10 positive and show nuclear labeling for β-catenin.
该肿瘤的鉴别诊断包括高分化胰腺内分泌肿瘤和腺泡细胞癌(下文讨论)。免疫组化非常有用,因为该肿瘤呈CD10阳性,并且核表达β-catenin。
假囊肿(Pseudocyst)
The definition of a pseudocyst is “lacking an epithelial lining.” This is a walled-off area of fat necrosis and granulation tissue containing high levels of pancreatic enzymes that is not usually mistaken for a malignancy, clinically or microscopically. Remember that most pseudocysts are actually extrapancreatic.
假囊肿的定义是“缺乏被覆上皮”。这是由脂肪坏死和肉芽组织构成的封闭区域,含有高水平的胰酶,在临床或显微镜下通常不会被误认为恶性肿瘤。记住,大多数假囊肿实际上是胰腺外的。
胰腺其他实性肿瘤(Other Solid Tumors in the Pancreas)
There are only two pancreatic cell types not yet discussed (not counting soft tissue elements such as vessels and nerves): the acinar cells (exocrine secretory) and the islet cells (endocrine). Neoplasms composed of these cells are important to remember because they can release enzymes or hormones, causing dramatic clinical presentations. These tumors can also show considerable histologic overlap and may require special stains to distinguish.
只有两种胰腺细胞类型尚未讨论(不包括软组织成分,如血管和神经等):腺泡细胞(外分泌细胞)和胰岛细胞(内分泌细胞)。要记住由这些细胞组成的肿瘤,因为它们可以释放酶或激素,引起剧烈的临床表现。这些肿瘤也可以表现出相当大的组织学重叠,可能需要特别的染色方法来区分。
腺泡细胞癌(Acinar Cell Carcinoma)
Acinar cell carcinomas are rare tumors of older adults, usually male. The usual appearance is that of nodules and sheets of densely packed amphophilic (purple) cells with uniform round nuclei. Growth may be trabecular, nested, or acinar (arranged around tiny lumens). Prominent nucleoli are often seen and are a clue to the diagnosis. Like the benign acinar cells, these tumors are usually positive for trypsin.
腺泡细胞癌是老年人发生的罕见肿瘤,通常为男性。通常表现为结节和成片的密集的双染性(紫色)细胞,核圆而一致。生长方式可以是小梁状、巢状或腺泡状(排列在微腔周围)。常见显著的核仁,是诊断的线索。正如良性腺泡细胞,这些肿瘤通常表达胰蛋白酶。
高分化胰腺内分泌肿瘤(Well-Differentiated Pancreatic Endocrine Neoplasm)
Well-differentiated pancreatic endocrine neoplasms are simply the neuroendocrine tumors of the pancreas, also known as islet cell tumors. These tumors are usually well circumscribed and cellular, and the neoplastic cells tend to form nests or trabeculae. The cytology is that of a carcinoid (Figure 10.12). Some are functional and produce clinically significant levels of insulin, glucagon, somatostatin, or other peptides. Cytology is not an indication of behavior, usually, and a bland tumor may look just as bland when you later find it in the liver. Highgrade neuroendocrine tumors are rare in the pancreas and when present are usually of the small cell variety. Well-differentiated pancreatic endocrine neoplasms express neuroendocrine markers (SYN, CHR, CD56) as well as any peptides they may be producing.
高分化胰腺内分泌肿瘤是指胰腺的神经内分泌肿瘤,也称为胰岛细胞瘤。这些肿瘤通常边界清楚,细胞丰富,肿瘤细胞倾向于形成巢或小梁。细胞学表现为类癌(图10.12)。有些是功能性的,产生临床上显著水平的胰岛素、胰高血糖素、生长抑素或其他肽。细胞异型性不能提示生物学行为,形态温和的肿瘤也可能发生肝转移。胰腺高级别神经内分泌肿瘤很少见,通常为小细胞癌。高分化胰腺内分泌肿瘤表达神经内分泌标记物(SYN、CHR、CD56)以及它们可能产生的任何肽。
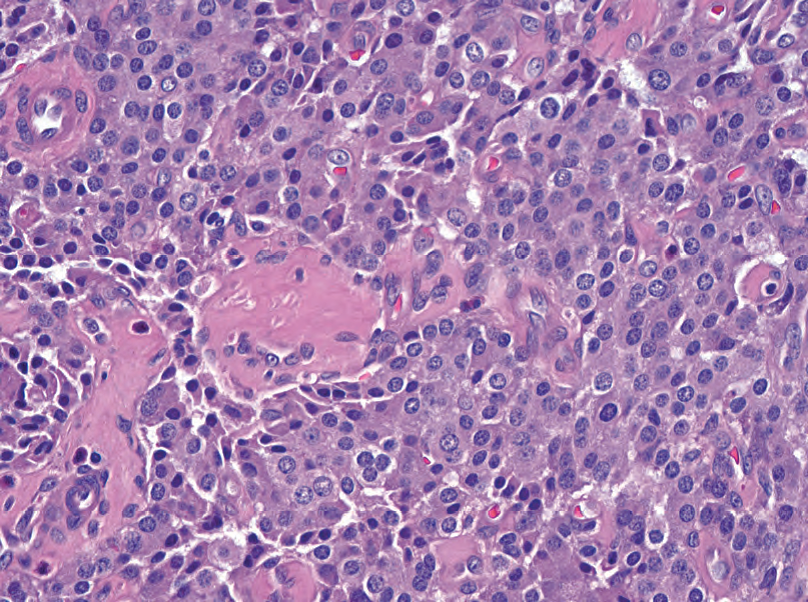
Figure 10.12. Islet cell tumor. This resembles carcinoids in other body sites, with round, well-spaced nuclei and speckled neuroendocrine-type chromatin.
图10.12.胰岛细胞瘤。类似于身体其他部位的类癌,具有圆形、分布均匀的核,和点彩状神经内分泌型染色质。
来源:
The Practice of Surgical Pathology:A Beginner’s Guide to the Diagnostic Process
外科病理学实践:诊断过程的初学者指南
Diana Weedman Molavi, MD, PhD
Sinai Hospital, Baltimore, Maryland
ISBN: 978-0-387-74485-8 e-ISBN: 978-0-387-74486-5
Library of Congress Control Number: 2007932936
© 2008 Springer Science+Business Media, LLC
仅供学习交流,不得用于其他任何途径。如有侵权,请联系删除。
本站欢迎原创文章投稿,来稿一经采用稿酬从优,投稿邮箱tougao@ipathology.com.cn
相关阅读
 数据加载中
数据加载中
我要评论

热点导读
-

淋巴瘤诊断中CD30检测那些事(五)
强子 华夏病理2022-06-02 -
【以例学病】肺结节状淋巴组织增生
华夏病理 华夏病理2022-05-31 -

这不是演习-一例穿刺活检的艰难诊断路
强子 华夏病理2022-05-26 -

黏液性血性胸水一例技术处理及诊断经验分享
华夏病理 华夏病理2022-05-25 -

中老年女性,怎么突发喘气困难?低度恶性纤维/肌纤维母细胞性肉瘤一例
华夏病理 华夏病理2022-05-07







共0条评论