外科病理学实践:诊断过程的初学者指南 | 第9章 肝
第9章 肝(Liver)
Liver biopsies are usually needle core biopsies. The most common reasons for a biopsy include monitoring disease progression in hepatitis, evaluating a transplanted patient for rejection or graft-versus-host disease (GVHD), and occasionally ruling out an additional disease process. Liver biopsies may also be done to diagnose a radiographic mass. At many institutions, all liver biopsy specimens are stained with trichrome (to evaluate fibrosis) or with an iron stain (to reveal abnormal iron in the tissue).
肝活检通常是针芯活检。活检最常见的原因包括监测肝炎的疾病进展,评估移植患者的排斥反应或移植物抗宿主病(GVHD),偶尔排除其他疾病过程。肝活检也可以用来诊断放射学肿块。在许多机构,所有肝活检标本都用三色染色(评估纤维化)或铁染色(显示组织中的异常铁)。
(译注:core needle biopsy,粗针穿刺活检。大号穿刺针带有内芯,粗针穿刺获取组织条,针芯把组织条推出来。而细针穿刺(FNA)获取细胞样本,一般没有组织结构,除非做细胞蜡块转换成组织标本)
解剖学(Anatomy)
Blood comes in via the portal vein (from the gut) and the hepatic artery (from the aorta). These vessels ramify into, eventually, the arteries and veins within the portal tracts. Blood gets from the terminal portal vessels to the outgoing central veins via the sinusoids—the long channels lined by hepatocytes. Once in the central vein, blood exits the liver via the left and right hepatic veins, which join the inferior vena cava (joining blood from the lower extremities).
血液通过门静脉(来自肠道)和肝动脉(来自主动脉)进入。这些血管最终分支成汇管区内的动脉和静脉。血液从终末门脉经过被覆肝细胞的长通道——肝窦,现经过中央静脉流出。一旦进入中央静脉,血液通过左肝静脉和右肝静脉流出肝,再进入下腔静脉(收集来自下肢的血液)。
Bile is created by the hepatocytes and exits into the bile canaliculi, which eventually coalesce into ductules and ducts in the portal tracts. These exit the liver via the hepatic ducts, which join the cystic duct (from the gallbladder) to enter the duodenum as the distal common bile duct.
胆汁由肝细胞产生并进入胆小管,最终汇入小导管和汇管区的导管。它们通过肝管离开肝,加入胆囊管(来自胆囊),通过远端胆总管进入十二指肠。
正常组织学(Normal Histology)
The liver is composed of three main components – the hepatocytes, the biliary system, and the vessels. Hepatocytes are large pink polygonal cells with dense round nuclei. Nucleoli, and occasional binucleate cells, are okay. The hepatocytes are organized into plates that are one hepatocyte thick and lined by reticulin. Between these plates are the sinusoids for blood. Running perpendicular to the sinusoids, and essentially invisible to light microscopy, are the bile canaliculi: tiny intercellular channels between the hepatocytes.
肝有三种主要成分:肝细胞、胆道系统和血管。肝细胞是大的粉红色多边形细胞,有密集的圆核。核仁,偶见双核细胞,都是正常现象。肝细胞组成一层细胞厚度的板状,被覆网状纤维。板块之间是血窦。胆小管垂直于肝窦,光镜基本上看不见,是肝细胞之间微小的细胞间通道。
Bile from the canaliculi makes its way to the bile ducts. The bile ducts are tubular structures with a low cuboidal epithelium (Figure 9.1). They are found in the portal tracts, which also contain branches of the hepatic artery and portal vein. These three components are also called the portal triad. Blood in both vessels is flowing into the liver; bile is flowing out. The portal tract also contains a small amount of connective tissue, which makes it stand out on a trichrome stain. The hepatocytes immediately surrounding the portal tract are called the limiting plate. The portal tract is usually the hotspot for inflammatory processes in the liver and so is important to identify on biopsy.
胆汁从胆小管进入胆管。胆管被覆低立方上皮(图9.1)。它们见于汇管区,汇管区也包含肝动脉和门静脉的分支。三者也称为门脉三联征。两条血管中的血液都流入肝;而胆汁流出肝。汇管区也含有少量结缔组织,三色染色很明显。紧邻汇管区的肝细胞称为限制板(界板)。汇管区通常是肝的炎症病变的热点区域,因此活检识别汇管区很重要。
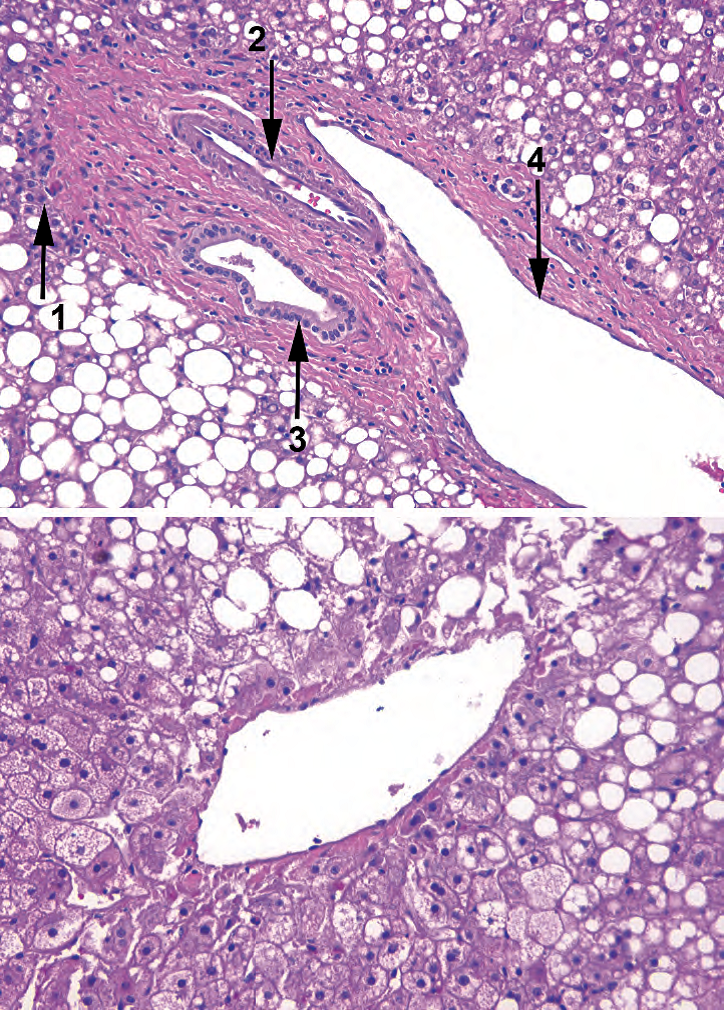
Figure 9.1. Portal tract and central vein. The upper panel shows a typical portal tract surrounded by the limiting plate of hepatocytes (1) and containing a branch of the hepatic artery (2), bile ductule (3), and portal vein (4). The lower panel shows a central vein from the same liver. Both panels show extensive steatosis.
图9.1。汇管区和中央静脉。上图为典型的汇管区,由肝细胞界板(1)包围,并包含肝动脉(2)、胆管(3)和门静脉(4)的分支。下图为同一肝的中央静脉。两图均显示广泛的脂肪变性。
The third vessel in the liver unit is the central vein or terminal venule. This is a thin-walled vessel surrounded by hepatocytes and nothing else (see Figure 9.1). It contains blood on its way out of the liver.
肝单位的第三条血管是中央静脉或终末小静脉。这是一个薄壁血管,周围环绕着肝细胞,没有其他东西(见图9.1)。它含有从肝流出的血液。
The lobule is an architectural unit with the central vein as its center and portal tracts at the periphery. Centrilobular refers to a process involving the central vein. This is the most easily visualized anatomic unit (Figure 9.2).
小叶是以中央静脉为中心,周围为汇管区的结构单位。小叶中心性是指累及中央静脉的病变。这是最容易识别的解剖单位(图9.2)。
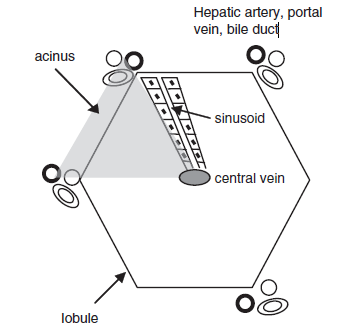
Figure 9.2. Liver organization. The acinus is a triangular, physiologic unit, while the lobule is a hexagonal anatomically based unit.
图9.2。肝组织结构模式图。腺泡是三角形的生理单位,小叶六边形的解剖单位。
图中文字:
lobule 小叶
acinus 腺泡
Hepatic artery, portal vein, bile duct 肝动脉、门静脉和胆管
sinusoid 窦
central vein 中央静脉
The acinus is an architectural unit with the portal tract at the base (as the source of blood flow) and the central vein at the tip. In this model, the area closest to the source of blood and oxygen is zone 1, and the most peripheral cells are zone 3. Ischemia and toxic insults affect the zones differentially. This is more of a physiologic unit and the one used when describing liver findings.
腺泡是一个结构单位,底部为汇管区(供血),尖端为中央静脉。在这个模型中,距离血和氧的来源最近的区域是1区,最外围的细胞是3区。缺血和毒性损伤对这些区域有不同的影响。这更像生理单位,用于描述肝的形态学表现。
非肿瘤性或炎症性疾病的分类(Nonneoplastic or Inflammatory Disease Categories)
It is helpful to think of the different liver compartments separately, because histologic findings can often be grouped as well:
把不同的肝成分分开考虑是有帮助的,因为组织学检查结果通常也可以分组:
Diseases of hepatocytes: the viral hepatitides, autoimmune hepatitis, steatohepatitis and alcoholic disease, and drug toxicity
肝细胞疾病:病毒性肝炎、自身免疫性肝炎、脂肪性肝炎和酒精性疾病以及药物中毒
Diseases of the biliary system: autoimmune biliary diseases (primary sclerosing cholangitis and primary biliary cirrhosis), obstruction, atresia, transplant rejection, GVHD, and drugs
胆道系统疾病:自身免疫性胆道疾病(原发性硬化性胆管炎和原发性胆汁性肝硬化)、梗阻、闭锁、移植排斥、GVHD和药物
Diseases of the vasculature: transplant rejection, GVHD, and systemic vasculitides
血管系统疾病:移植排斥、GVHD和系统性血管炎
The portal tract represents a collision of all three compartments. Therefore, inflammation of the portal tract is found in all of these diseases.
汇管区代表所有三种成分的碰撞。因此,所有这些疾病都发现了汇管区炎症。
病理学表现(Pathologic Findings)
The liver has only so many ways to respond to an insult or injury. An acute injury in the liver looks similar to that in any other organ: widespread edema, acute and chronic inflammation, and/or necrosis. Subacute or chronic injury generally has mainly mononuclear inflammatory cells as well as individual cell necrosis or degeneration. The final result of chronic injury from many causes is cirrhosis, or end-stage liver disease. Therefore, many diseases in the liver have histologic overlap, and, in the case of cirrhosis, often you cannot tell what the original disease process was. For this reason, the most important skill in interpreting the liver biopsy is recognizing injury to the different compartments. Attaching a diagnosis to this collection of findings requires clinical information. The most common patterns of injury are the following:
肝对冒犯或损伤的反应方式只有这么多。肝的急性损伤与任何其他器官的损伤相似:广泛水肿、急性和慢性炎症,和/或坏死。亚急性或慢性损伤通常主要有单个核炎症细胞以及单个细胞坏死或变性。多种原因造成的慢性损伤最终导致肝硬化或终末期肝病。因此,肝的许多疾病都有组织学上的重叠,在肝硬化的情况下,通常无法判断最初的疾病过程是什么。因此,解释肝活检最重要的技巧是识别不同成分的损伤。综合这些表现,加上临床信息,形成诊断。损伤的最常见模式如下:
肝细胞成分(Hepatocellular compartment)
Portal inflammation: Inflammatory cells are present within the portal tract. In chronic hepatitis and autoimmune disorders, the infiltrate is predominantly mononuclear.
汇管区炎症:汇管区存在炎症细胞。慢性肝炎和自身免疫性疾病主要是单个核细胞浸润。
Interface activity (periportal hepatitis, piecemeal necrosis): Inflammation, usually lymphocytic, occurs in the limiting plate. This looks like portal inflammation spilling out into the hepatocytes (Figure 9.3). Note that the word activity when describing something in the liver does not mean neutrophils.
界面活动性炎症(汇管区周围肝炎,碎片性坏死):炎症,通常为淋巴细胞性,发生在界板内。这看似汇管区炎症扩散到肝细胞(图9.3)。注意,当描述肝的某些东西时,“活动”一词并不意味着中性粒细胞。
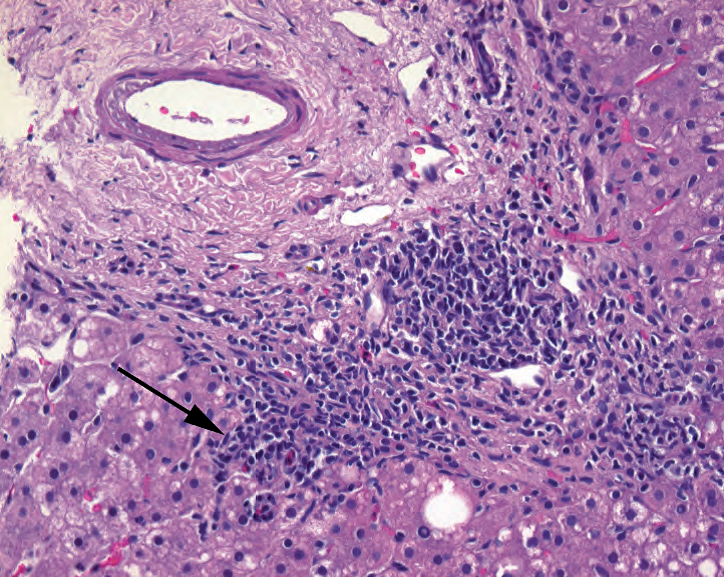
Figure 9.3. Portal inflammation. This is an example of chronic viral hepatitis. Lymphocytes in the portal tract spill out into the limiting plate of surrounding hepatocytes (arrow).
图9.3。汇管区炎症。这例是慢性病毒性肝炎。汇管区中的淋巴细胞外溢到周围界板中的肝细胞(箭号)。
Lobular inflammation: Inflammation, usually chronic, and/or necrosis of the hepatocytes are at a distance from the portal tracts. Also called spotty necrosis, this appears as little clusters of lymphocytes destroying individual hepatocytes out in the lobules. Do not count lymphocytes in the sinuses, which are physiologic.
小叶炎症:炎症(通常为慢性)和/或肝细胞坏死位于距离汇管区较远的位置。也称为斑点状坏死,表现为小叶中的小簇淋巴细胞破坏单个肝细胞。不要计数肝窦中的淋巴细胞,这是生理现象。
Vacuolar degeneration (balloon cell change): This is one way in which hepatocytes can die. The cytoplasm swells and becomes feathery and pale.
空泡变性(气球细胞改变):这是肝细胞可能死亡的一种方式。细胞质膨胀,变成羽毛状和淡染。
Acidophilic bodies: This is another way in which hepatocytes die. These cells are similar to dyskeratotic cells in the skin; they are bright pink, rounded up, with pyknotic nuclei.
嗜酸小体:这是肝细胞死亡的另一种方式。这些细胞类似于皮肤中的角化不良细胞;它们呈明亮的粉红色,变圆,核固缩。
°Fibrosis: Fibrosis is a general term indicating too much collagen. Fibrosis begins as an increase in collagen around the portal tract (portal fibrosis), and eventually spreads to connect adjacent portal tracts or central veins by thin webs of collagen (bridging fibrosis). The end stage of the process is cirrhosis, which is the division of the liver into individual nodules separated by thick bands of fibrosis (Figure 9.4).
纤维化:纤维化是指胶原蛋白过多的总称。纤维化开始于汇管区周围胶原的增多(汇管区纤维化),最终通过胶原的细网扩散到相邻的汇管区或中央静脉(桥接纤维化)。该过程的终末期是肝硬化,肝被分成由粗束纤维化分隔的单个结节(图9.4)。
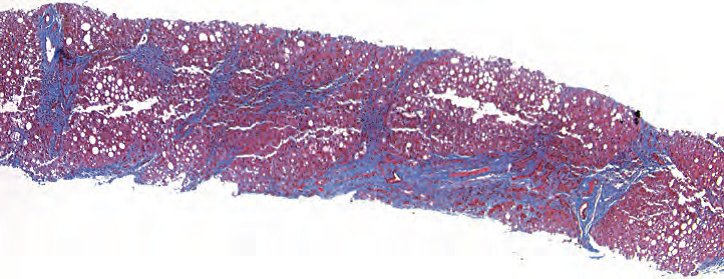
Figure 9.4. Cirrhosis in a biopsy specimen. In this trichrome stain, collagen is blue, while hepatic parenchyma is red. Collagen can be seen outlining the lobules of the liver, bridging the portal tracts and creating a nodular pattern.
图9.4。活检标本中的肝硬化。三色染色,胶原呈蓝色,而肝实质呈红色。可见胶原勾勒出肝小叶,桥接汇管区,形成结节状结构。
Steatosis: Steatosis literally means fat in the hepatocytes. Steatosis can be physiologic in small amounts (< 5%), but 30%–60% involvement is considered moderate steatosis. Over 60% involvement is marked or severe disease. Macrovesicular steatosis means large single vacuoles in each hepatocyte and is typical of fatty liver, alcoholic disease, and nonalcoholic steatohepatitis. Pure microvesicular steatosis looks like foamy cytoplasm and is characteristic of mitochondrial injury such as in Reye’s syndrome.
脂肪变性:脂肪变性字面上是指肝细胞中的脂肪。少量的脂肪变性可能是生理性的(<5%),但一般认为,30%-60%受累是中度脂肪变性,60%以上受累是明显或严重的疾病。大泡性脂肪变性是指每个肝细胞中存在单个大空泡,是脂肪肝、酒精性疾病和非酒精性脂肪性肝炎的典型表现。单纯的微囊性脂肪变性看似泡沫状细胞质,是线粒体损伤的特征,如Reye综合征。
Steatohepatitis: Steatohepatitis is steatosis plus inflammation or injury. Neutrophils are not necessary for the diagnosis, but some evidence of hepatocyte injury is (Figure 9.5). This includes lobular inflammation, hepatocyte necrosis, pericellular fibrosis, balloon cells, and Mallory’s hyaline (see below).
脂肪性肝炎:脂肪性肝炎是脂肪变性加上炎症或损伤。中性粒细胞不是诊断所必需的,但肝细胞损伤的一些证据是必需的(图9.5)。这些证据包括小叶炎症、肝细胞坏死、细胞周围纤维化、气球细胞和Mallory透明小体(见下文)。
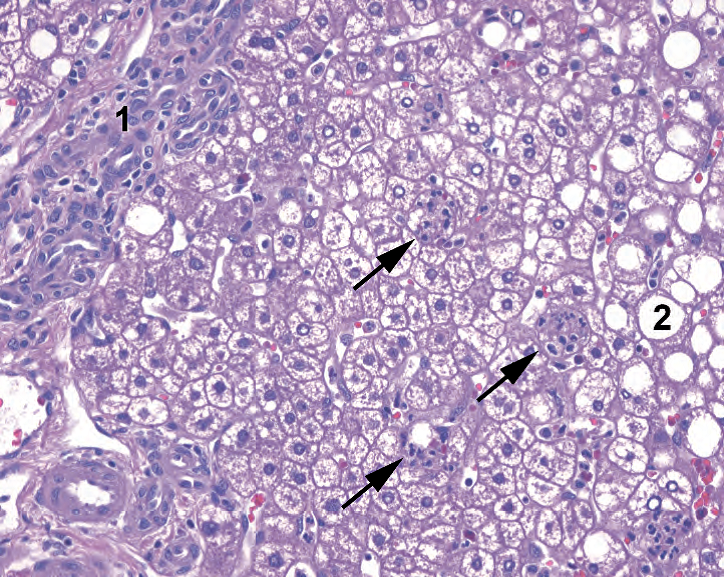
Figure 9.5. Steatohepatitis. An adjacent portal tract (1) shows minimal inflammation. In the lobule, there is macrovesicular steatosis (2) and collections of neutrophils attacking individual hepatocytes (arrows).
图9.5。脂肪性肝炎。邻近的汇管区(1)显示轻微炎症。在小叶中,有大泡性脂肪变性(2)和攻击单个肝细胞的中性粒细胞聚集(箭头)。
Mallory’s hyaline (Mallory bodies): Mallory bodies are irregular worm-like pink blobs of condensed cytoskeleton in the cytoplasm, especially within balloon cells (Figure 9.6). They are associated with steatohepatitis, especially alcoholic disease.
Mallory透明小体(Mallory小体):Mallory小体是细胞质中不规则的蠕虫状粉红色凝缩细胞骨架,尤其是在气球细胞内(图9.6)。它们与脂肪性肝炎,尤其是酒精性疾病有关。
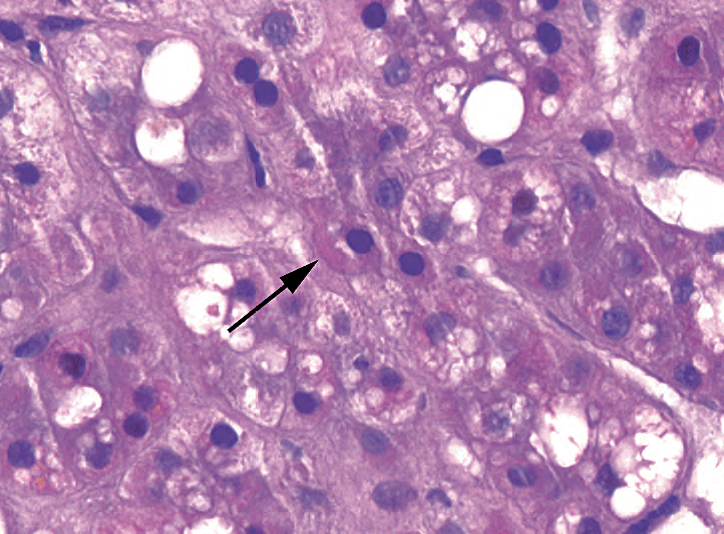
Figure 9.6. Mallory’s hyaline. In the background of steatosis and inflammation, a pink refractile wormlike structure in the hepatocyte (arrow) is evidence of cytoskeletal collapse.
图9.6。Mallory透明小体。在脂肪变性和炎症的背景下,肝细胞中的粉红色折光性蠕虫状结构(箭头),是细胞骨架塌陷的证据。
Megamitochondria: Megamitochondria are markedly enlarged mitochondria, which look like red blood cells entrapped in the hepatocyte cytoplasm.
巨线粒体:巨线粒体是明显增大的线粒体,看起来像陷入肝细胞质中的红细胞。
Iron accumulation: Abnormal levels of iron are detected with either hematoxylin and eosin or iron stain. If severe, iron accumulation may indicate hemochromatosis or be secondary to other hepatocellular processes.
铁累积:HE染色或铁染色检测到铁含量异常。如果严重,铁积累可能提示血色病或继发于其他肝细胞病变。
胆成分(Biliary compartment)
Cholestasis: Cholestasis is the backup of bile in the liver. This may be caused by ext rahepatic obstruction to flow, intrahepatic biliary disease, or impaired excretion by the hepatocytes themselves.
胆汁淤积症:胆汁淤积症是胆汁在肝中蓄积。这可能是由于肝外血流受阻、肝内胆道疾病或肝细胞自身排泄受损所致。
Bile duct proliferation: An increase in the number of bile duct profiles occurs in each portal tract; on average, there should be one to two per tract. Many of the new ductules are small, peripheral, and poorly formed. Bile duct proliferation occurs as a response to obstruction to flow. Other findings in obstruction include visible bile in hepatocytes or canaliculi, edema and inflammation (especially acute) in the portal tracts, eventually ductular atrophy, and finally widespread fibrosis (Figure 9.7).
胆管增殖:每个汇管区中胆管的数量增加;每个汇管区平均应该有一到两个。许多新生小导管是小的、位于周边的、形状不完好的。胆管增殖是对胆汁流出受阻的反应。梗阻的其他表现包括肝细胞或胆小管中可见胆汁、汇管区水肿和炎症(特别是急性炎症),最终导管萎缩,广泛纤维化(图9.7)。
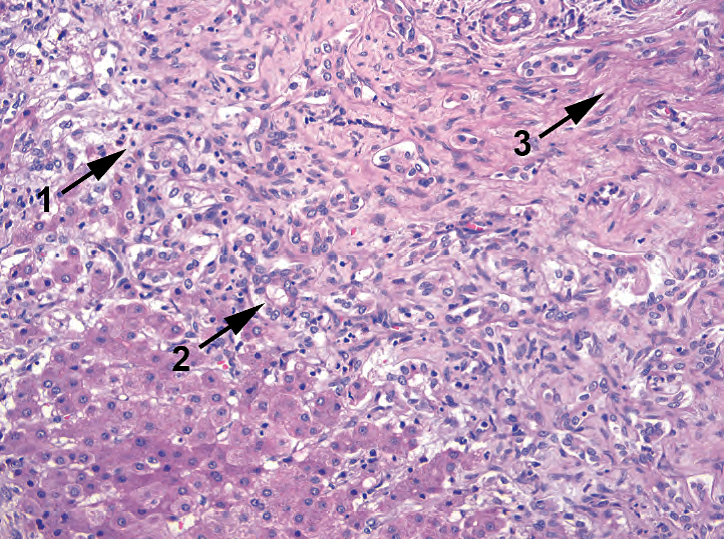
Figure 9.7. Bile stasis. In this example of congenital biliary atresia, the downstream obstruction to flow creates the triad of acute inflammation (1), a proliferation of poorly formed bile ductules (2), and the accumulation of golden globs of bile (not seen here). This will progress to fibrosis (3) and eventually loss of ductules.
图9.7。胆汁淤积。这例是先天性胆道闭锁。胆汁流出的下游受阻,导致三联征:急性炎症(1)、形状不完好的小胆管增殖(2)和金黄色胆汁小球堆积(此处未看到)。这将进展为纤维化(3)并最终导致小导管消失。
Bile duct injury: Bile duct injury is identified by lymphocytes in the bile duct epithelium and vacuolar degeneration or dropout of the epithelial cells. The end stage is ductopenia. Injury to the bile ducts can indicate a biliary disease, such as autoimmune (primary biliary cirrhosis) or rejection. Bile duct injury is usually patchy, so multiple portal tracts must be examined.
胆管损伤:胆管损伤表现为胆管上皮中出现淋巴细胞、上皮细胞空泡变性或脱落。终末期为导管减少症。胆管损伤可能提示胆道疾病,如自身免疫性(原发性胆汁性肝硬化)或排斥反应。胆管损伤通常呈斑片状,因此必须检查多个汇管区。
Ductopenia: Ductopenia is loss of bile ducts, an indicator of chronic damage to the biliary system. Recognizing ductopenia, a diagnosis of absence, requires a conscious effort to look for bile ducts. Finding a bile duct in less than 80% of the portal tracts is abnormal.
胆管减少:胆管减少是胆管丢失,提示胆道系统的慢性损伤。胆管减少是通过发现胆管减少或消失而诊断的,需要有意识地努力寻找胆管。在不到80%的汇管区发现胆管是不正常的。
血管成分(Vascular compartment)
Venulitis (endothelitis): Venulitis is damage to the endothelium of the portal or central veins by inflammatory cells. It is usually an indication of rejection or GVHD.
静脉炎(内皮炎):静脉炎是炎症细胞对门静脉或对中央静脉内皮的损害。通常提示排斥反应或GVHD。
Extramedullary hematopoiesis: Hematopoietic precursors (megakaryocytes are the most distinctive) are present in the liver sinuses. It is generally an indication of bone marrow disease (but is physiologic in fetuses and infants).
髓外造血:肝窦中存在造血细胞前体(巨核细胞是最独特的)。它通常提示骨髓疾病(但在胎儿和婴儿是生理性的)。
慢性肝炎(Chronic Hepatitis)
Biopsies in chronic hepatitis, generally hepatitis C, are done to track disease progression, with the ultimate endpoint being cirrhosis. Sign out of a hepatitis biopsy specimen should include three key prognostic factors: etiology (if known), grade (degree of inflammation and necrosis), and stage (degree of fibrosis), plus any other disease process present (such as steatohepatitis).
慢性肝炎(通常是丙型肝炎)的活检是为了追踪疾病进展,最终终点是肝硬化。肝炎活检标本的签发应包括三个关键的预后因素:病因(如果知道)、分级(炎症和坏死程度)、分期(纤维化程度),以及任何其他存在的疾病过程(如脂肪性肝炎)。
There are many different scoring systems used to quantify grade and stage, as all clinicians love a number. However, most numeric scoring systems can be translated to or from adjectives, which convey the same information (for example, scores 0 to 4 corresponding to none, minimal, mild, moderate, or severe inflammation; and none, portal, periportal, bridging, or cirrhotic fibrosis). Calibrating these levels takes some experience, and thresholds may vary by institution.
有许多不同的评分系统用于量化分级和分期,因为所有临床医生都喜欢一个数字。然而,大多数数字评分系统可以翻译成或来自形容词,形容词传达相同的信息(例如,0到4分对应无、轻微、轻度、中度或重度炎症;无、汇管区、汇管区周围、桥接或肝硬化纤维化)。准确掌握这些水平需要一些经验,并且阈值可能因机构而异。
The changes in viral hepatitis are nonspecific. The differential includes most other hepatocellular processes.
病毒性肝炎的改变没有特异性。鉴别诊断包括大多数其他肝细胞病变。
排斥反应或移植物抗宿主病的移植活检(Transplant Biopsy for Rejection or Graft-Versus-Host Disease)
The changes seen in cellular rejection and GVHD are histologically similar; one occurs in the setting of a liver transplant and the other in a bone marrow transplant. Both are divided into acute and chronic. (Hyperacute rejection implies an antibody response and is rare and immediate, not usually diagnosed by biopsy.)
细胞排斥反应和GVHD的改变在组织学上相似;一种发生在肝移植,另一种发生在骨髓移植。两者都分为急性和慢性。(超急性排斥反应意味着抗体反应,罕见且立即发生,通常不通过活检诊断。)
Acute cellular rejection: Acute rejection usually occurs 5–30 days after transplant, but can be longer. Changes include the following:
急性细胞排斥反应:急性排斥反应通常发生在移植后5-30天,但可能更长。改变包括:
Mixed portal tract inflammation, including lymphocytes, neutrophils, and eosinophils
混合性汇管区炎症,包括淋巴细胞、中性粒细胞和嗜酸性粒细胞
Venulitis
静脉炎
Bile duct inflammation and damage (Figure 9.8)
胆管炎症和损伤(图9.8)
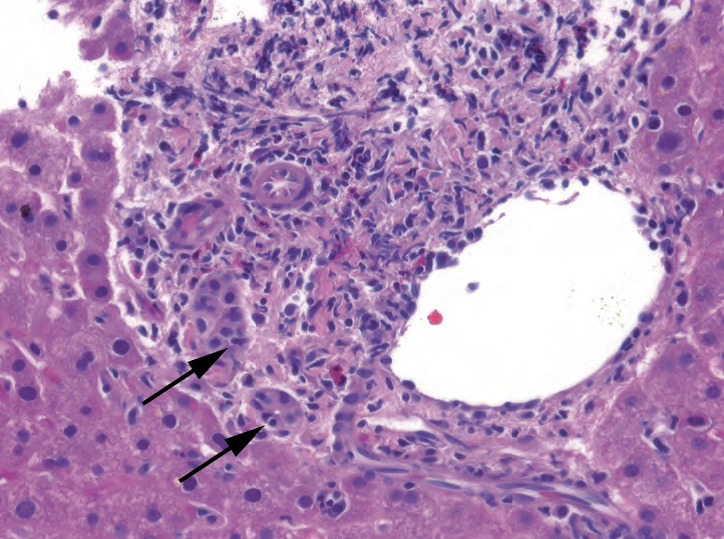
Figure 9.8. Acute rejection. Acute rejection refers to the attack on the bile ducts and venules by lymphocytes, which are seen invading the duct epithelium (arrows).
图9.8。急性排斥反应。急性排斥反应是指胆管和小静脉受到攻击,可见淋巴细胞侵入胆管上皮(箭号)。
Chronic rejection: Chronic rejection usually occurs after more than 1 year. Changes are primarily those of ductopenia and fibrosis.
慢性排斥反应:慢性排斥通常发生在1年以上。改变主要是导管减少和纤维化。
Note that the changes of rejection must be differentiated from recurrent hepatitis C, an inevitable occurrence in patients who lost their first liver to hepatitis C (occurs from 3 to 9 months after transplantation).
注意,排斥反应的改变必须与复发性丙型肝炎区分开来,丙型肝炎导致第一肝丧失的患者不可避免地会发生排斥反应(发生在移植后3至9个月)。
原发性胆汁性肝硬化和原发性硬化性胆管炎(Primary Biliary Cirrhosis and Primary Sclerosing Cholangitis)
Primary biliary cirrhosis and primary sclerosing cholangitis are hard to keep straight. The bullet version is as follows:
原发性胆汁性肝硬化和原发性硬化性胆管炎很难掌握。要点如下:
Primary biliary cirrhosis (occurs much more often in women than in men):
原发性胆汁性肝硬化(女性比男性更常见):
Primary biliary cirrhosis is a chronic destructive intrahepatic cholangitis (inflammation of the intrahepatic bile ducts).
原发性胆汁性肝硬化是一种慢性破坏性肝内胆管炎(肝内胆管的炎症)。
Cirrhosis is an end-stage feature.
肝硬化是终末期特征。
It is associated with antimitochondrial antibody.
与抗线粒体抗体相关。
Findings are nonspecific and patchy but include inflammation and injury to the bile ducts, especially granulomatous, followed by proliferation and cholestasis, then eventually ductopenia and cirrhosis (Figure 9.9).
病理表现没有特异性,斑片状,但包括胆管炎症和损伤,尤其是肉芽肿,随后是增殖和胆汁淤积,最后是导管减少和肝硬化(图9.9)。
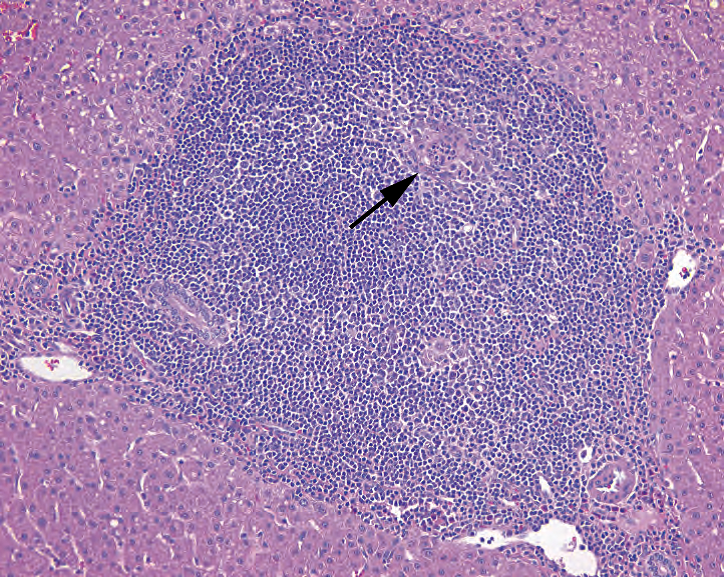
Figure 9.9. Primary biliary cirrhosis. There is a granulomatous inflammation of the portal tract, with destruction of a bile ductule (arrow).
图9.9。原发性胆汁性肝硬化。汇管区有肉芽肿性炎症,伴有小胆管破坏(箭号)。
The etiology is direct damage to bile duct epithelium.
病因是直接损伤胆管上皮。
Primary sclerosing cholangitis (occurs more often in men than in women):
原发性硬化性胆管炎(男性多于女性):
Primary sclerosing cholangitis is an inflammatory disease of the extrahepatic (and large intrahepatic) ducts.
原发性硬化性胆管炎是肝外(和肝内大)导管的炎症性疾病。
It leads to patchy stricturing lesions, visible on cholangiogram.
导致胆管斑片状狭窄性病变,造影可见。
It is associated with inflammatory bowel disease and p-ANCA.
与炎症性肠病和p-ANCA相关。
The histologic picture is also nonspecific but dominated by ductular proliferation and cholestasis.
组织学表现也没有特异性,但主要是导管增殖和胆汁淤积。
The etiology is unknown but may be a fibrotic process of the connective tissue surrounding the bile ducts, causing secondary stricture and damage.
病因不明,但可能是胆管周围结缔组织的纤维化过程,导致继发性狭窄和损伤。
肿块性病变(肿瘤)(Mass Lesions (Neoplasms))
The most common cause of mass lesions in the liver is metastatic disease. However, there are primary lesions of all three components of the liver: hepatocytes, biliary epithelium, and vessels. Within each category, it can be difficult to differentiate neoplastic from nonneoplastic, and benign from malignant, on resection, let alone on biopsy. However, here is a brief list of features that favor one over the other.
肝肿块最常见的原因是转移性疾病。然而,肝的三个组成部分(肝细胞、胆管上皮和血管)都有原发性病变。在每一组的病变中,在切除时都很难区分肿瘤性和非肿瘤性,以及良性和恶性,更不用说活检了。然而,这里有一个简单的功能列表,其中一个优于另一个。
肝细胞(Hepatocellular)
Focal nodular hyperplasia: Focal nodular hyperplasia is essentially an island of cirrhosis occurring in the background of a noncirrhotic liver. This is not a clonal process, so there is more than one cell type; in addition to hepatocytes are bile ducts and fibrous septae. There is no capsule but sometimes a central scar. The lesion is composed of nodules divided by bands of fibrosis and thick vessels.
局灶性结节性增生:局灶性结节性增生本质上是发生在非肝硬化背景下的肝硬化孤岛。这不是克隆性病变,所以有不止一种细胞类型;除肝细胞外,还有胆管和纤维间隔。没有包膜,但有时有中央瘢痕。病变由结节组成,结节由纤维束和厚壁血管分隔。
Adenoma: Adenomas are benign clonal neoplasms. They occur mainly in noncirrhotic livers of adult women taking oral contraceptive pills. Adenomas are circumscribed, partially encapsulated masses of uniform, bland-looking hepatocytes with no central veins or bile ducts (although there are diffuse prominent arterioles). The cells may be pale due to steatosis or glycogen or discolored with bile (which has no place to go). When visualized with reticulin stain, the hepatocyte plates are still only one to two cells thick (every cell touches reticulin).
腺瘤:腺瘤是良性克隆性肿瘤。主要发生在服用口服避孕药的成年妇女的非肝硬化肝。腺瘤表现为边界清楚的、部分包裹肿块,肝细胞均匀分布,形态学温和,没有中央静脉或胆管(尽管有弥漫分布的明显小动脉)。细胞可能因脂肪变性或糖原而淡染,或因胆汁而变色(胆汁无处可去)。当用网状纤维染色观察时,肝板仍然只有一到两层细胞厚度(每个细胞都接触网状纤维,译注,或网状纤维包围单个细胞)。
Well-differentiated hepatocellular carcinoma: Well-differentiated hepatocellular carcinoma (HCC) can be very difficult to distinguish from an adenoma histologically. However, HCC generally occurs in the setting of cirrhosis, unlike the adenoma. As with an adenoma, there are no bile ducts or central veins, and you may see intracellular bile (Figure 9.10). Nuclei may be large, hyperchromatic, and irregular. A reticulin stain shows a breakdown in architecture, and plates may be three or more cells in thickness.
高分化肝细胞癌:高分化肝细胞癌(HCC)在组织学上很难与腺瘤区分。然而,与腺瘤不同,肝癌通常发生于肝硬化。与腺瘤一样,没有胆管或中央静脉,可以看到细胞内胆汁(图9.10)。核可能增大,深染,不规则。网状纤维染色显示结构破坏,肝板厚度可能≥3层细胞厚度。
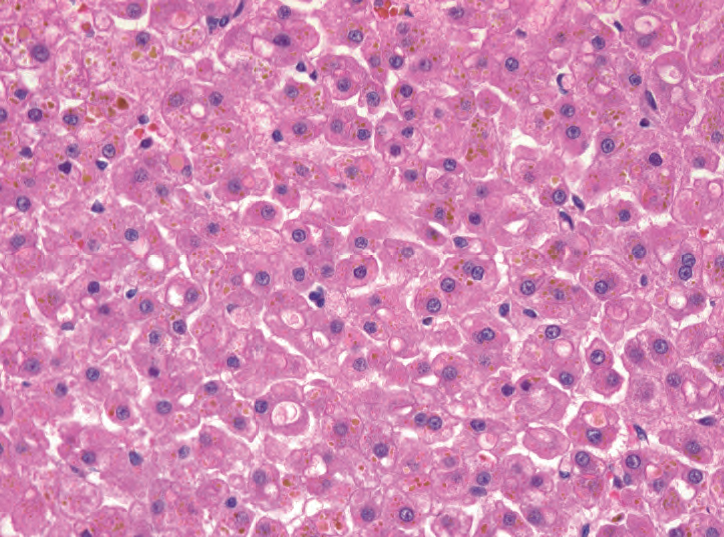
Figure 9.10. Well-differentiated hepatocellular carcinoma. Golden bile can be seen in the tumor cells, which are hard to differentiate from normal liver. Portal tracts are absent.
图9.10。高分化肝细胞癌。肿瘤细胞中可见金黄色胆汁,很难与正常肝区分。汇管区缺失。
Poorly differentiated hepatocellular carcinoma: Poorly differentiated HCC can be very pleomorphic and hard to identify as hepatic. The presence of bile, if any, is still a give away.
低分化肝细胞癌:低分化肝细胞癌可能是多形性显著,很难识别为肝细胞癌。胆汁的存在,如果有的话,仍然是诊断线索。
Fibrolamellar hepatocellular carcinoma: Fibrolamellar HCC is a variant of well-differentiated HCC occurring in children and young adults. It is typified by oncocytic cells with prominent nucleoli in a dense fibrotic stroma.
纤维板层肝细胞癌:纤维板层HCC是发生于儿童和年轻成人的一种变异型高分化HCC。它的典型特征是在致密的纤维化间质中有显著核仁的嗜酸细胞。
胆(Biliary)
Bile duct adenoma: A bile duct adenoma is usually<1 cm and subcapsular (often sampled on frozen section), with a tangle of small simple tubules, with or without inflammation and fibrosis. It may produce mucin but not bile. Think of this as a benign biliary epithelial neoplasm (Figure 9.11).
胆管腺瘤:胆管腺瘤通常<1 cm且为被膜下(通常在冰冻切片上取样),伴简单小管的缠结,伴或不伴炎症和纤维化。它可能产生黏液,但不产生胆汁。将其视为良性胆管上皮肿瘤(图9.11)。
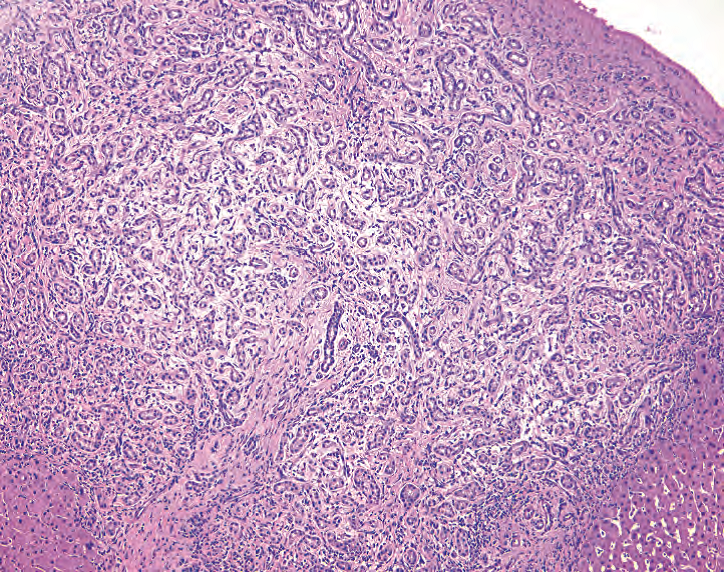
Figure 9.11. Bile duct adenoma. This is a benign tangle of proliferating bile ducts, surrounded by edema, which may mimic desmoplasia. Bile is absent, as is any cytologic atypia.
图9.11。胆管腺瘤。良性增殖性胆管的缠结,胆管周围有水肿,可能貌似促结缔组织增生反应。胆汁不存在,任何程度的细胞学异型性也不存在。
Bile duct hamartoma: Also called von Meyenburg complex, a bile duct hamartoma is also usually < 1 cm and subcapsular (often sampled on frozen section). However, it generally shows more dilated and angular tubules in a loose connective tissue stroma and often produces bile. Think of this as a disordered reduplication of the portal tract. The consequences of confusing the hamartoma with the adenoma are minimal.
胆管错构瘤:也称为von Meyenburg复合体,胆管错构瘤通常也是<1 cm且位于包膜下(通常在冰冻切片上取样)。然而,它通常显示出更扩张的和成角的小管,位于疏松的结缔组织间质中,并且经常产生胆汁。将其视为汇管区的无序重复。将错构瘤与腺瘤混淆的后果很轻微。
Cholangiocarcinoma: Cholangiocarcinoma is a primary malignancy of the bile ducts that appears as a nondescript adenocarcinoma infiltrating the liver. There is no definitive way to distinguish it from a metastatic lesion except by history. Although bile is not present in a cholangiocarcinoma, mucin is common, as is an intense desmoplastic response (Figure 9.12).
胆管癌:胆管癌是胆管的原发性恶性肿瘤,表现为浸润肝的非特殊腺癌。除了病史外,没有明确的方法将其与转移性病变区分开来。尽管胆管癌不存在胆汁,但常见黏液,也常见强烈的促结缔组织增生反应(图9.12)。
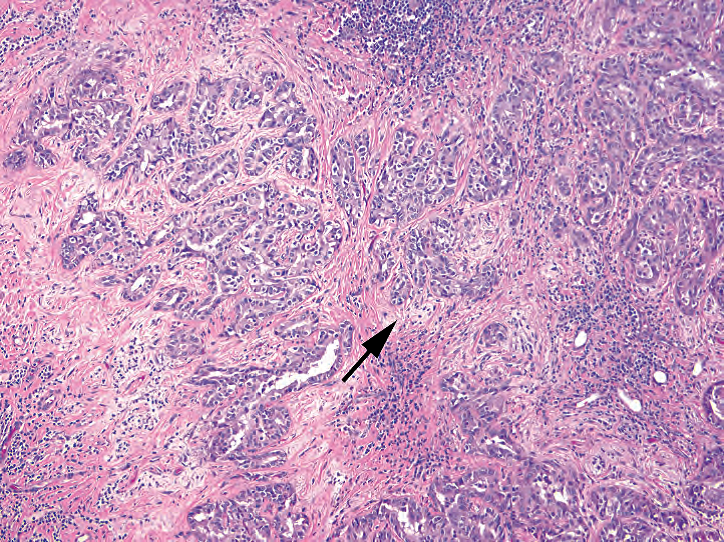
Figure 9.12. Cholangiocarcinoma. A nondescript adenocarcinoma, cholangiocarcinoma produces an intense desmoplastic response in the stroma (arrow; the pale swirling fibrosis surrounding the malignant glands).
图9.12。胆管癌。胆管癌是一种非特殊腺癌,在间质中产生强烈的促结缔组织增生反应(箭号;围绕恶性腺体的淡染漩涡状纤维化)。
Vascular lesions
Cavernous hemangiomas are benign vascular lesions.
海绵状血管瘤是良性血管病变。
Epithelioid hemangioendotheliomas have a low malignant potential.
上皮样血管内皮瘤具有低度恶性潜能。
Angiosarcomas are malignant.
血管肉瘤是恶性。
来源:
The Practice of Surgical Pathology:A Beginner’s Guide to the Diagnostic Process
外科病理学实践:诊断过程的初学者指南
Diana Weedman Molavi, MD, PhD
Sinai Hospital, Baltimore, Maryland
ISBN: 978-0-387-74485-8 e-ISBN: 978-0-387-74486-5
Library of Congress Control Number: 2007932936
© 2008 Springer Science+Business Media, LLC
仅供学习交流,不得用于其他任何途径。如有侵权,请联系删除。
本站欢迎原创文章投稿,来稿一经采用稿酬从优,投稿邮箱tougao@ipathology.com.cn
相关阅读
 数据加载中
数据加载中
我要评论

热点导读
-

淋巴瘤诊断中CD30检测那些事(五)
强子 华夏病理2022-06-02 -
【以例学病】肺结节状淋巴组织增生
华夏病理 华夏病理2022-05-31 -

这不是演习-一例穿刺活检的艰难诊断路
强子 华夏病理2022-05-26 -

黏液性血性胸水一例技术处理及诊断经验分享
华夏病理 华夏病理2022-05-25 -

中老年女性,怎么突发喘气困难?低度恶性纤维/肌纤维母细胞性肉瘤一例
华夏病理 华夏病理2022-05-07







共0条评论