外科病理学实践:诊断过程的初学者指南 | 第7章 胃和十二指肠
第7章 胃和十二指肠(Stomach and Duodenum)
胃(Stomach)
The stomach is composed of several anatomic zones. Moving proximal to distal, like a piece of food, you pass (1) the gastroesophageal junction, (2) the cardia, (3) the fundus or body, (4) the antrum, and (5) the pylorus (Table 7.1 and Figure 7.1). For the pathologist, there are essentially two types of mucosa in the stomach (Figure 7.2), antral and oxyntic, as cardiac mucosa looks very similar to antrum. The entire stomach epithelium is composed of pits (invaginations from the surface) and glands (deep to the pits). The surface and pits are lined by columnar mucinous epithelium (sometimes called foveolar type) which stains bright pink with PAS/AB. The regions of the stomach are divided by the type of underlying glands:
胃有几个解剖区域。食物从近端到远端移动,经过(1)胃食管交界处,(2)贲门,(3)胃底或胃体,(4)胃窦,(5)幽门(表7.1和图7.1)。对于病理医生来说,胃基本上有两种类型的粘膜(图7.2),胃窦粘膜和泌酸粘膜,因为贲门黏膜与胃窦非常相似。整个胃上皮由小凹(表面的内陷)和腺体(深至小凹下方)组成。表面和小凹被覆柱状粘液上皮(有时称为小凹型),PAS/AB染色呈明亮的粉红色。胃的区域按以下腺体的类型划分:

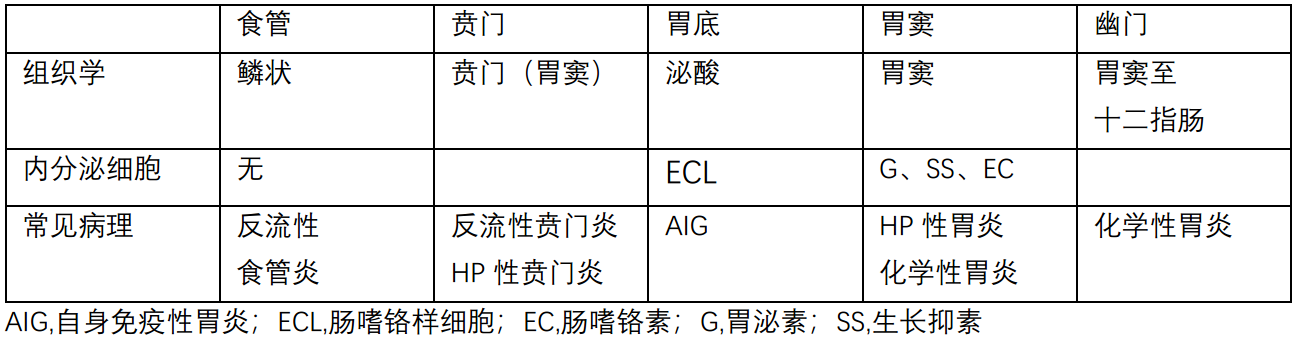
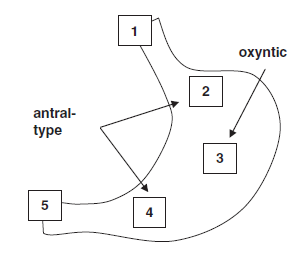
Figure 7.1. Localization of anatomic regions within the stomach: (1) the gastroesophageal junction, (2) the cardia, (3) the fundus, (4) the antrum, and (5) the pylorus. Antral-type (mucinous) mucosa is seen in the cardia and the antrum.
图7.1.胃内解剖区域的定位:(1)胃食管交界处,(2)贲门,(3)胃底,(4)胃窦,(5)幽门。胃窦型(粘液性)粘膜见于贲门和胃窦。
(图中文字:antral-type,胃窦型;oxyntic,泌酸)
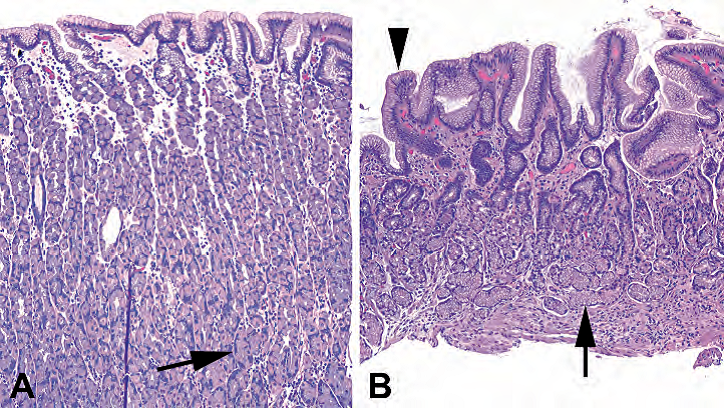
Figure 7.2. Antral and oxyntic mucosa. (A) Oxyntic mucosa is relatively thick, with most of the mucosa occupied by secretory cells (arrow), the parietal and chief cells. The surface is composed of mucinous foveolar epithelium. (B) Antral mucosa is thinner, and the glands are mucinous instead of secretory (arrow). However, the surface is still composed of foveolar epithelium (arrowhead).
图7.2.胃窦粘膜和泌酸粘膜。(A)泌酸粘膜相对较厚,大部分粘膜被分泌细胞(箭号)、壁细胞和主细胞占据。表面由粘液性小凹上皮组成。(B)胃窦粘膜较薄,腺体为粘液性而非分泌性(箭号)。然而,表面仍然由小凹上皮(箭头)组成。
Antral mucosa (found in the antrum): The glands are loosely packed, mucinous, and occupy about half of the epithelial thickness. Cardiac mucosa looks very similar.
胃窦粘膜(见于胃窦):腺体松散排列,粘液性,占上皮厚度的一半左右。贲门黏膜看起来很相似。
Oxyntic mucosa (found in the fundus or body): The glands are tightly packed, contain granular parietal (pink, acid-secreting) and chief (purple, enzyme-secreting) cells, and occupy three fourths of the mucosal thickness.
泌酸粘膜(见于胃底或胃体):腺体紧密排列,含有颗粒状壁细胞(粉红色,分泌酸)和主细胞(紫色,分泌酶),占粘膜厚度的四分之三。
Transitional mucosa: Features of both antral and oxyntic are present. Transitional mucosa represents the overlap zone.
移行粘膜:兼有胃窦粘膜和泌酸粘膜的特征。移行粘膜代表重叠区。
It is important to note what kind of epithelium is present in the biopsy tissue so that the endoscopists can correlate with what they saw. Also, there are certain processes that differentially affect mucosal types; clarifying the type of epithelium involved may change the differential.
重要的是要注意活检组织中存在什么类型的上皮,以便内镜医生能够结合胃镜下所见。此外,某些疾病对不同的粘膜类型有不同的影响;识别所累及的上皮类型可能会改变鉴别诊断。
Endocrine cells occur singly in the glands. In the body, they are mainly enterochromaffinlike cells, while in the antrum they are mixed gastrin, enterochromaffin, and somatostatin. A chromogranin stain highlights all endocrine cells. A gastrin stain should be positive only in the antrum.
内分泌细胞单独出现在腺体中。胃体主要是肠嗜铬样细胞(ECL),而在胃窦,它们是混合胃泌素(G)、肠嗜铬素(EC)和生长抑素(SS)的细胞。嗜铬素染色突出显示所有内分泌细胞。胃泌素染色应当仅在胃窦呈阳性。
活检的观察方法(The Approach to the Biopsy)
Survey the glandular epithelium at low power:
低倍镜下观察腺上皮:
The first thing to notice is what kind of mucosa you have and whether it correlates with what the endoscopist told you. Then decide on the color of the biopsy specimen. A healthy stomach is a pretty pale pink, overall. If your general impression is blue, this probably indicates inflammation in the stroma, such as in gastritis (Figure 7.3). If your impression is that of a pink stroma with unusually dark and distinct glands, you may be looking at chemical gastritis.
首先要注意,你看到什么类型的粘膜,是否符合内镜医生所述。然后判断活检标本的颜色。总体上,健康的胃是一种相当淡的粉红色。如果你看到总体印象是蓝色,可能提示间质中有炎症,如胃炎(图7.3)。如果你的印象是粉红色的间质,伴有不同寻常的深染的明显的腺体,那么可能是化学性胃炎。
Goblet cells are usually visible from low power, especially on the PAS/alcian blue (AB) stain (as indigo-blue, bulbous cells), and indicate intestinal metaplasia, a marker of chronic irritation in the stomach. Remember that true goblet cells are usually interspersed among nonmucinous pink cells (absorptive). A row of back-to-back tall mucinous cells, even if blue on PAS/AB, is unlikely to be actual intestinal metaplasia.
杯状细胞通常在低倍镜下可见,尤其是在PAS/AB染色(呈靛蓝色、球状细胞),提示肠化生,这是胃慢性刺激的标志。请记住,真正的杯状细胞通常散布在非粘液性粉红色细胞(吸收细胞)中。一排背靠背的高柱状粘液细胞,即使在PAS/AB上呈蓝色,也不太可能是真正的肠化生。
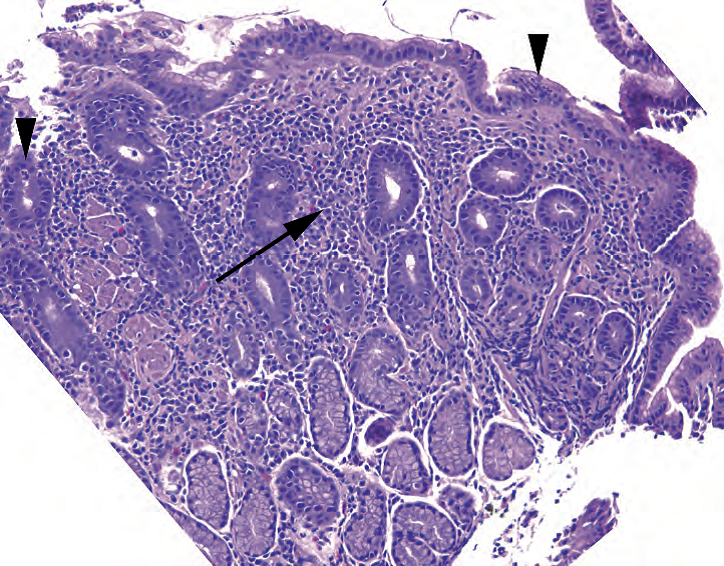
Figure 7.3. Helicobacter pylori gastritis, antrum. In this disease, the low-power impression is that of a “blue” biopsy due to the dense inflammatory infiltrate in the lamina propria (arrow). There are lymphocytes, plasma cells, and neutrophils. Neutrophils in the glandular or surface epithelium (arrowheads) indicate an active component to this gastritis. Helicobacter pylori organisms are pictured in Chapter 3.
图7.3.幽门螺杆菌(HP)性胃炎,胃窦。低倍印象是个“蓝色”活检,因为固有层有密集的炎性浸润(箭号)。有淋巴细胞、浆细胞和中性粒细胞。腺上皮或表面上皮有中性粒细胞(箭头)提示该胃炎的活动性成分。HP病原体图像见第3章。
Areas of exudate, neutrophils, debris, and ragged-looking glands indicate an erosion or ulcer. Ulcers are discussed in more detail later.
渗出物、中性粒细胞、碎屑和破损的腺体区域提示糜烂或溃疡。溃疡将在后面进行更详细的讨论。
在高倍镜下,评估炎症:
On higher power, assess the inflammation:
A few lymphocytes, plasma cells, and eosinophils are okay in the stomach, especially in the antrum, where there is more space between glands. However, back-to-back lymphocytes and plasma cells pushing aside or crowding the glands indicate a chronic gastritis.
胃内有少量淋巴细胞、浆细胞和嗜酸性粒细胞是正常的,尤其是胃窦,腺体之间距离较宽。然而,背靠背的淋巴细胞和浆细胞、将腺体推开或或使腺体变得拥挤,提示慢性胃炎。
The presence of neutrophils in the stomach indicates activity and is not called acute as in other organs. If you have only mononuclear cells, you have an inactive chronic gastritis, but if there are any neutrophils embedded in the surface or glandular epithelium, you have an active chronic gastritis.
胃中出现中性粒细胞提示活动性炎症,不像其他器官那样称为急性炎症。如果你只看到单个核的炎症细胞,称为非活动性慢性胃炎,但是如果表面或腺上皮中有任何数量的中性粒细胞,称为活动性慢性胃炎。
Neutrophils, chronic gastritis, and lymphoid follicles are all associated with Helicobacter pylori infection. The tiny rods are visible on hematoxylin and eosin stain (see Chapter 3) but are better seen on Diff-Quik or Giemsa. They should be visible at 40× as tiny discrete seagull-shaped rods in the pit lumens or on the surface, mainly in the antrum, unless there is intestinal metaplasia, a hostile mucosa for these bugs. If you have no significant inflammation, do not work too hard looking for H. pylori.
中性粒细胞、慢性胃炎和淋巴滤泡都与HP感染有关。HE染色(见第3章)可以看到细小的杆状细菌,但Diff-Quik染色或Giemsa染色更明显。在40倍镜下应该可以看到HP,是小凹或表面的离散的细小海鸥形杆状细菌。HP主要分布在胃窦,除非有肠化生,肠化生粘膜不利于细菌生存。如果没有明显的炎症,不要太努力寻找HP。
How many lymphocytes does it take to diagnose lymphoma? Lymphoma in the stomach often arises from mucosa-associated lymphoid tissue (MALT), is of marginal zone phenotype, and is a result of chronic H. pylori infection. You should see sheets of monocytoid B cells (fried egg–like) and lymphoepithelial lesions (lymphocytes embedded in epithelium) before considering this diagnosis.
诊断淋巴瘤需要多少淋巴细胞?胃淋巴瘤通常起源于粘膜相关淋巴组织(MALT),属于边缘区表型,是慢性HP感染的结果。在考虑这个诊断之前,你应该能看到成片的单核细胞样B细胞(煎蛋样)和淋巴上皮病变(嵌入上皮的淋巴细胞)。
小凹增生(Foveolar Hyperplasia)
Especially in the antrum, the stomach is vulnerable to bile reflux. Bile and other sources of chemical irritation, such as nonsteroidal anti-inflammatory drugs, cause a process called foveolar hyperplasia. The surface mucin cells proliferate, giving the surface a papillary appearance and the pits a corkscrew profile. The mucinous cells lose mucin, and the cytoplasm becomes more dark or opaque; the nuclei also may become hyperchromatic, adding to the dark look (Figure 7.4). Smooth muscle fibers proliferate and can be seen stranding up between the pits. Inflammation in the stroma is not a prominent feature, so the lamina propria is often fairly pale, even edematous. This appearance is called chemical gastritis, and it is very common.
尤其是在胃窦,胃粘膜容易受到胆汁反流的损伤。胆汁和其他化学刺激源,如非甾体抗炎药,会导致小凹增生。表面黏液细胞增生增殖,使表面呈乳头状,小凹呈螺旋状。黏液细胞失去黏液,细胞质变得更深染或不透明;细胞核也可能变得深染,总体上看起来颜色更深(图7.4)。平滑肌纤维增殖,可见其在小凹之间向上延伸。间质炎症不明显,因此固有层通常相当淡染,甚至水肿。这种表现称为化学性胃炎,很常见。
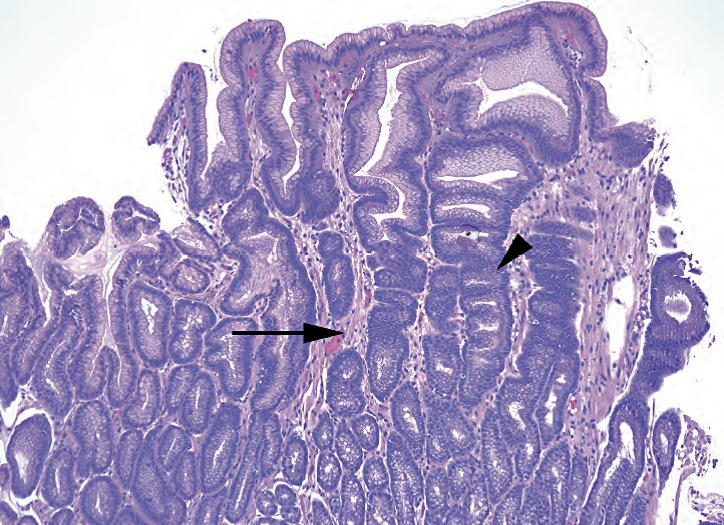
Figure 7.4. Chemical gastritis, antrum. In chemical gastritis, the lamina propria shows very little inflammation, unlike in Helicobacter pylori gastritis. The lamina propria is pale and sometimes edematous such that the dark reactive nuclei of the glands stand out sharply in contrast. The corkscrew profile of the hyperplastic glands is a second classic feature (arrowhead), as is the presence of thin strands of smooth muscle between the glands (arrow).
图7.4.化学性胃炎,胃窦。与HP性胃炎不同,化学性胃炎的固有层只能很轻微的炎症。固有层淡染,有时水肿,腺体的深染核与之形成鲜明对比。增生腺体呈螺旋状轮廓,是第二个典型特征(箭头),腺体之间存在细股平滑肌(箭号)。
萎缩(Atrophy)
The loss of glands in the stomach, in any region, is called atrophy. Atrophy can be difficult to assess on any given biopsy specimen, as badly oriented sections, a healing ulcer, or dense inflammation can all lead to the appearance of loss of glands. Regardless of the cause, true atrophy, as an end-stage response to severe chronic damage, should be accompanied by intestinal metaplasia and inflammation (Figure 7.5). The two principal types of atrophy are the following:
在胃的任何部位,腺体的缺失都称为萎缩。在某个具体的活检标本上可能难以评估萎缩,因为包埋方向不对、愈合性溃疡或密集炎症都可能导致腺体丢失。无论病因如何,作为严重慢性损伤的终末期反应,真正的萎缩应伴有肠化生和炎症(图7.5)。萎缩的两种主要类型如下:
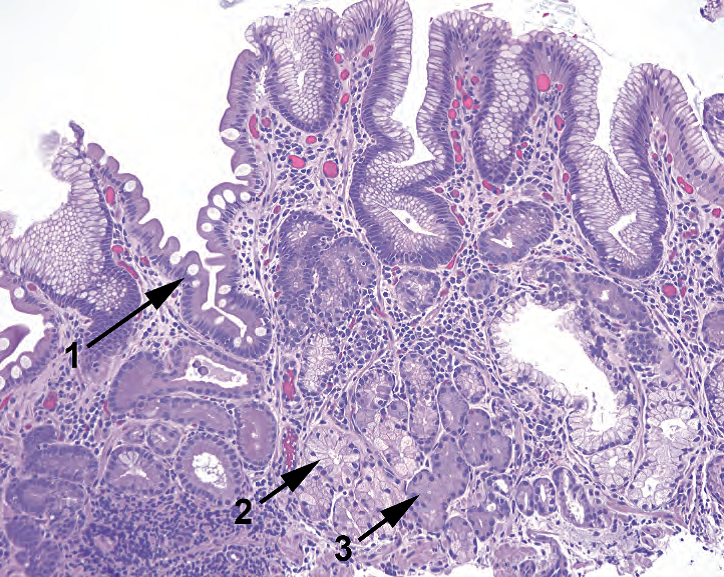
Figure 7.5. Autoimmune gastritis. This fundic biopsy specimen shows several features of atrophy. The surface shows goblet cells, which are indicative of intestinal metaplasia (1); deep to this there is inflammation and replacement of the secretory glands by mucinous, antral-type glands (2). Some residual oxyntic cells are also visible (3).
图7.5.自身免疫性胃炎。胃底活检标本显示萎缩的几个特征。表面显示杯状细胞,提示肠化生(1);深层是炎症,由粘液腺、胃窦腺取代了分泌腺(2)。也可见一些残余的泌酸细胞(3)。
Helicobacter pylori gastritis (formerly known as environmental metaplastic atrophic gastritis) is secondary to H. pylori infection, primarily affects the antrum, and involves loss of glands in the setting of active chronic gastritis and intestinal metaplasia. You may also see lymphoid follicles and pit abscesses (pits full of neutrophils).
HP性胃炎(旧称为环境化生萎缩性胃炎):继发于HP感染,主要影响胃窦,在活动性慢性胃炎和肠化生的背景中显示腺体缺失。你也可能看到淋巴滤泡和小凹脓肿(小凹充满中性粒细胞)。
Autoimmune gastritis (formerly known as autoimmune metaplastic atrophic gastritis), a result of the autoimmune destruction of the parietal cells in the fundus, shows loss of fundic glands in the setting of chronic inflammation and intestinal metaplasia. It is associated with a compensatory antral G-cell hyperplasia and hypergastrinemia. The hyperplasia may even progress to microcarcinoids or tumorlets. In autoimmune gastritis, you should not see activity or lymphoid follicles.
自身免疫性胃炎(AIG,旧称为自身免疫性化生萎缩性胃炎):胃底壁细胞自身免疫性破坏的结果,在慢性炎症和肠化生的背景中显示胃底腺体缺失。它伴随代偿性胃窦G细胞增生和高胃泌素血症。增生甚至可能进展为微小癌或微肿瘤。在自身免疫性胃炎,你应该看不到活动性炎症或淋巴滤泡。
In severe autoimmune gastritis, the body of the stomach comes to resemble the antrum due to the atrophy of the secretory glands. It can therefore be difficult to decide if it is antrum or truly atrophic oxyntic mucosa. A gastrin stain, which will stain only true antrum, will help.
在严重的自身免疫性胃炎中,由于分泌腺萎缩,胃体变得象胃窦。因此,难以确定是胃窦还是真正萎缩的泌酸粘膜。胃泌素染色,只会染色真正的胃窦,会有所帮助。
淋巴瘤(Lymphoma)
Although diffuse large B-cell lymphomas are the most common lymphoma in the stomach, and should be considered if you see sheets of very ugly cells, differentiating them from normal inflammation is not usually a problem. Low-grade lymphomas, however, are tricky, especially given that most arise in the setting of chronic H. pylori gastritis. As mentioned earlier, you may see a background of lymphoepithelial lesions, which are collections of lymphocytes that appear to be eating glands (Figure 7.6), lymphoid follicles, and neoplastic plasma cells. Mucosa-associated lymphoid tissue lymphoma is usually of the marginal zone type, which is monocytoid in appearance (small round nuclei surrounded by a halo of clear cytoplasm).
尽管弥漫大B细胞淋巴瘤(DLBCL)是胃中最常见的淋巴瘤,如果你看到成片的非常丑陋的细胞,它们与正常炎症的区分通常不成问题,那么你就应该考虑DLBCL。然而,低级别淋巴瘤是棘手的问题,特别是考虑到它们大多数发生在慢性HP性胃炎的背景下。如前所述,你可能会看到淋巴上皮病变、淋巴滤泡和肿瘤性浆细胞的背景。淋巴上皮病变是淋巴细胞的聚集,似乎在吃掉腺体(图7.6)。粘膜相关淋巴组织淋巴瘤(MALToma)通常为边缘区型,肿瘤细胞呈单核细胞样(小而圆的核被透明的细胞质空晕包围)。
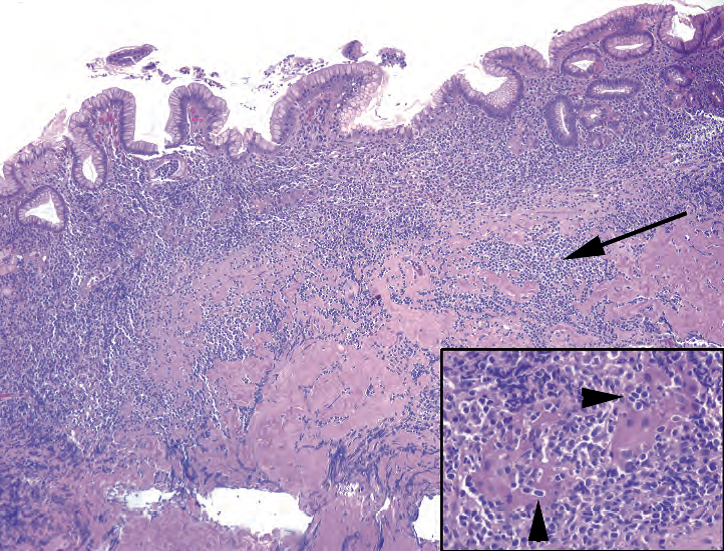
Figure 7.6. Mucosa-associated lymphoid tissue lymphoma. There are sheets of lymphocytes under the epithelium and dissecting into the muscularis mucosa (arrow). Inset: Lymphoepithelial lesions are typical, in which residual glands (seen here as little more than islands of pink cytoplasm) are infiltrated and destroyed by lymphocytes (arrowheads).
图7.6.MALToma。上皮下方有成片的淋巴细胞,切割并进入粘膜肌层(箭号)。插图:典型的淋巴上皮病变,残留的腺体(这里看到的只是粉红色的细胞质岛)被淋巴细胞浸润和破坏(箭头)。
Immunostains are often used to establish the diagnosis. In a MALT lymphoma, the majority of the cells should be B cells (CD20+) that also stain for CD43. Normal T cells may also stain for CD43, so you must mentally subtract out the background T cells (shown by CD3 stain). Helpfully, in chronic gastritis, most of the lymphocytes are T cells.
免疫染色通常用于确定诊断。在MALToma中,大多数细胞应为B细胞(CD20+)并且也显示CD43+。正常T细胞也是CD43+,所以你必须在头脑中减去背景T细胞(CD3染色显示)。在慢性胃炎中,大多数淋巴细胞是T细胞,这有助于鉴别。
溃疡(Ulcers)
An ulcer is a full-thickness defect of the epithelium down to muscularis mucosa (an erosion is more superficial). It is accompanied by fibrinopurulent exudate and/or granulation tissue, plus reparative glands. Search the periphery for the reason for the ulcer, including H. pylori, chemical gastritis, and adjacent cancer. Reparative glands appear as small, angulated glands with little mucin, and the lamina propria around them may be fibrotic. This can be difficult to distinguish from invasive carcinoma. However, reparative glands should have small or reactive nuclei and should have an overall streaming parallel arrangement, as they all want to orient to the surface (Figure 7.7).
溃疡是上皮至粘膜肌层的全层缺损(糜烂更为表浅)。它伴有纤维素性渗出物和/或肉芽组织,以及修复性腺体。在周围寻找溃疡的原因,包括HP、化学性胃炎和邻近的癌症。修复腺表现为小的、成角的腺体,含有很少粘液,其周围固有层可能纤维化。这很难与浸润性癌区分。然而,修复腺应该有小的或反应性的细胞核,并且整体上应该呈流水状平行排列,因为它们都希望朝向表面(图7.7)。
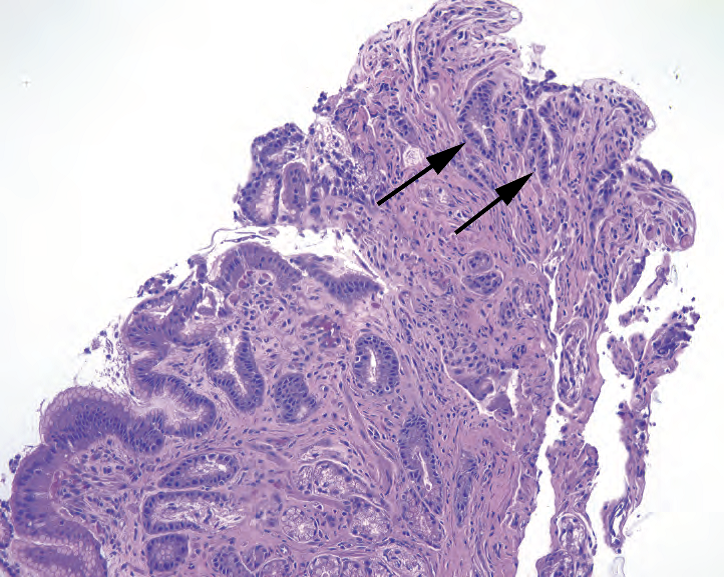
Figure 7.7. Reparative changes next to an ulcer. The tip of this fragment has a collection of poorly formed glands with an infiltrative look and minimal cytoplasm, giving the appearance of a high nuclear/ cytoplasmic ratio (arrows). However, the nuclei are of about the same size and shape as the rest of the gastric glands, and these small glands stream in parallel toward the surface, consistent with regenerative or reparative glands.
图7.7.溃疡旁边的修复性改变。该片段的一端有一团尚未完全成形的腺体,看起来像浸润,只有极少的细胞质,呈现较高的核质比(箭号)。然而,细胞核的大小和形状与其他胃腺体大致相同,这些小腺体呈流水状平行的朝向表面,符合再生性或修复性腺体。
息肉(Polyps)
A reasonable differential for a polypoid structure in the stomach includes the following:
胃的息肉样结构的合理鉴别诊断包括:
Fundic gland polyps: Fundic gland polyps look like oxyntic mucosa but with cystically dilated glands (Figure 7.8). They are common in older people. Multiple polyps occur in familial adenomatous polyposis.
胃底腺息肉:看起来像泌酸粘膜,但腺体囊性扩张(图7.8)。老年人常见。多发性息肉发生于家族性腺瘤性息肉病。
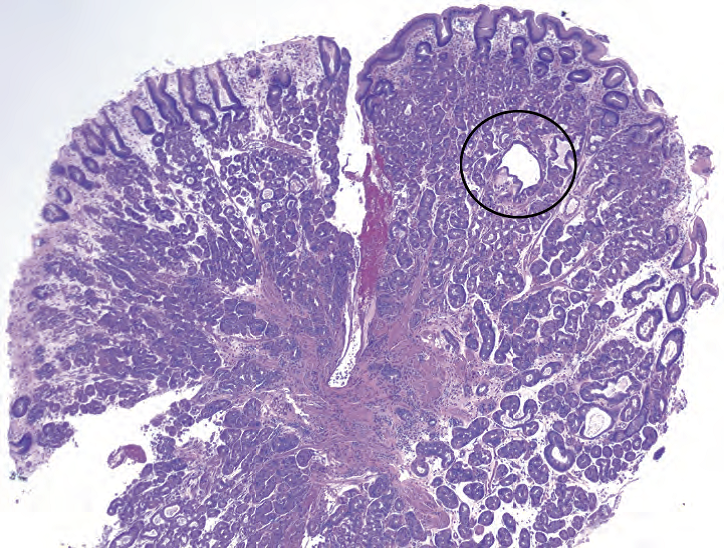
Figure 7.8. Fundic gland polyp. This polypoid fragment shows oxyntic- or fundic-type glands, with occasional dilated glands (circle).
图7.8.胃底腺息肉。息肉样片段显示有泌酸型或胃底型腺体,少数腺体扩张(圆圈)。
Hyperplastic polyps: Polyps are hyperplastic, elongated, or cystic foveolar pits with mild inflammation (Figure 7.9). They are usually associated with background gastritis.
增生性息肉:息肉含有增生性、拉长的或囊性小凹,伴有轻度炎症(图7.9)。它们常有胃炎背景。
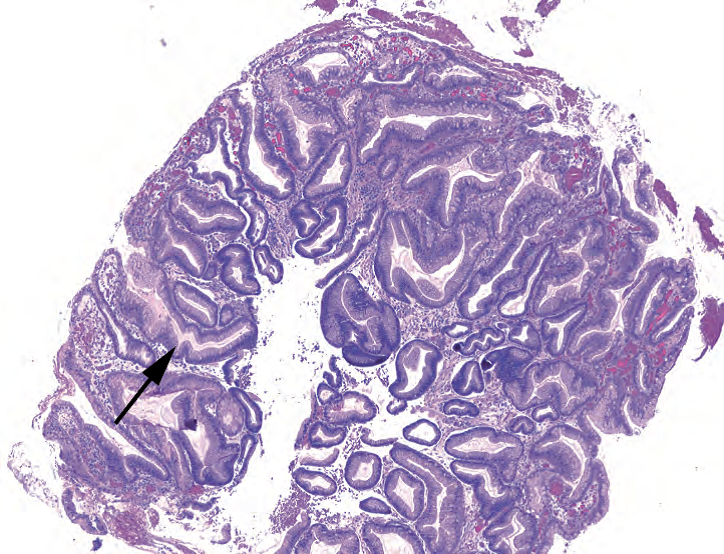
Figure 7.9. Hyperplastic polyp. This polyp is reminiscent of chemical gastritis, with corkscrew glands (arrow) and hyperplastic foveolar epithelium. Inflammation and intestinal metaplasia may be present.
图7.9.增生性息肉。这个息肉让人想起化学性胃炎,有螺旋状腺体(箭号)和增生的小凹上皮。可能存在炎症和肠化生。
Adenomas: These are neoplastic and dysplastic nodules that can be either gastric type or intestinal type. The gastric type is not associated with malignancy, but the intestinal type can be, such as a tubular adenoma.
腺瘤:这是一种肿瘤性和异型增生性结节,可以是胃型或肠型。胃型不伴有恶性肿瘤,但肠型可能伴有恶性肿瘤,如管状腺瘤。
异型增生对比癌(Dysplasia Versus Carcinoma)
Dysplasia in the stomach is assessed similarly to dysplasia in Barrett’s esophagus (see Chapter 6). As in Barrett’s esophagus, intestinal metaplasia is an abnormal finding but by itself is not dysplasia. Dysplasia in gastric mucosa initially begins to look like a tubular adenoma of the colon (it gets blue). The nuclei show increased hyperchromatism and pleomorphism, high nuclear/cytoplasmic ratio, loss of mucin vacuoles, crowding and pseudostratification, and loss of polarity. High-grade dysplasia tends to be called when invasive carcinoma is suspected but cannot be proven. Carcinoma in situ is not used in this situation; think of high-grade dysplasia as synonymous.
胃的异型增生的评估方法与Barrett食管的异型增生相似(见第6章)。正如Barrett食管,肠化生是一种异常发现,但其本身不是异型增生。胃粘膜的异型增生第一眼看起来像结肠管状腺瘤(总体上变成蓝色)。核深染性和多形性,高核/质比,胞质失去黏液空泡,细胞拥挤和假复层,以及极性丧失。当怀疑有浸润性癌但不能证实时,通常称为高级别异型增生。在这种情况不叫原位癌;将高度异型增生视为原位癌的同义词。
Invasive adenocarcinoma comes in two types in the stomach: intestinal type (which looks like colon cancer, hence the name) and diffuse. The intestinal type is fairly easy to spot; it is usually associated with atrophy and intestinal metaplasia. The diffuse type is the poorly differentiated, often signet-ring, infiltrative cancer that can creep through the entire stomach and cause linitus plastica. Signet-ring cell carcinoma gets its name from the single vacuolated cells with displaced and indented nuclei (Figure 7.10). They can look like foamy macrophages, and they can blend almost imperceptibly into the stroma. Every stomach biopsy specimen should get a once-over at high power, such as 20×, to scan the lamina propria for signet rings. When they are there, often you will see the first one and then realize there are hundreds of them.
浸润性胃腺癌有两种类型:肠型(看起来像结肠癌,因此得名)和弥漫型。肠型比较容易发现;通常伴有萎缩和肠化生。弥漫型是一种分化差、常有印戒细胞的浸润性癌,可蔓延至整个胃,并引起皮革胃。印戒细胞癌得名于含有单个大空泡的癌细胞,空泡将细胞核挤向一侧,核被挤压变扁(图7.10)。它们可能看起来像泡沫状巨噬细胞,几乎不知不觉地融入间质。每个胃活检标本都要在高倍(如20倍)镜下进行一次扫描,以观察固有层是否有印戒细胞。只要固有层存在印戒细胞,通常你会看到第一个,然后意识到有成百上千个。
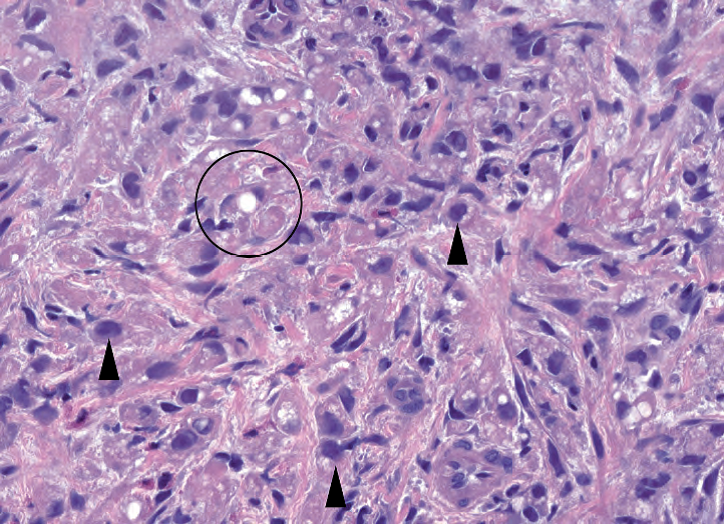
Figure 7.10. Signet-ring cell carcinoma. At low power, this sneaky tumor may be visible as little more than a slightly busy or cellular lamina propria. At high power, you can see individual signet-ring cells with single large mucin vacuoles (circle), plus other single infiltrating cells with large hyperchromatic nuclei (arrowheads). The signet-ring cells differ from fat cells by having large dark nuclei that protrude up from the surface of the central vacuole.
图7.10.印戒细胞癌。在低倍镜下,这种隐匿性肿瘤看起来可能只是固有层稍微杂乱、固有层细胞量稍微多一些。在高倍镜下,你可以看到单个印戒细胞,有单个大的黏液空泡(圆圈),以及其他单个浸润细胞伴大的深染核(箭头)。印戒细胞不同于脂肪细胞,印戒细胞有大的深染核,从中央空泡的表面鼓起来。
译者补充:2000年版WHO分类中胃印戒细胞癌
More than 50% of the tumour consists of isolated or small groups of malignant cells containing intracytoplasmic mucin. Superficially, cells lie scattered in the lamina propria, widening the distances between the pits and glands. The tumour cells have five morphologies: (1) Nuclei push against cell membranes creating a classical signet ring cell appearance due to an expanded, globoid, optically clear cytoplasm. These contain acid mucin and stain with Alcian blue at pH 2.5; (2) other diffuse carcinomas contain cells with central nuclei resembling histiocytes, and show little or no mitotic activity; (3) small, deeply eosinophilic cells with prominent, but minute, cytoplasmic granules containing neutral mucin; (4) small cells with little or no mucin, and (5) anaplastic cells with little or no mucin. These cell types intermingle with one another and constitute varying tumour proportions. Signet-ring cell tumours may also form lacy or delicate trabecular glandular patterns and they may display a zonal or solid arrangement.
印戒细胞癌中,50%以上的肿瘤是由含有胞质内粘液的孤立性或小团状恶性细胞组成。从表面上看(低倍镜下),细胞散在分布于固有层中,使得小凹和腺体之间的距离增宽。肿瘤细胞有五种形态:(1)典型印戒细胞:膨胀、球形、透明的细胞质,将核挤向细胞膜,形成典型的印戒细胞形态。它们含有酸性粘液,pH值2.5时AB染色阳性;(2)组织细胞样:其他弥漫性癌,含有类似组织细胞的癌细胞,核位于中央,核分裂活性轻微或无;(3)嗜酸细胞:小的强嗜酸性细胞,具有微小但显著的胞质颗粒,胞质颗粒含有中性粘液;(4)小细胞,黏液少或无;(5)间变细胞,黏液少或无。这些细胞类型相互混合,多少不等。印戒细胞癌也可形成花边状或纤细的小梁状腺样结构,可呈分区模式或实性排列。
粘膜下层(The Submucosa)
The submucosa is not always included in a biopsy specimen. It lies below the thin muscularis mucosa. However, there are some things that are more often found in the submucosa, including the following:
活检标本并不是都有粘膜下层。它位于薄的粘膜肌层下方。然而,粘膜下层有一些较常见的东西,包括:
Heterotopic pancreas is a nodule of well-developed pancreas.
异位胰腺,是发育良好的胰腺形成的结节。
Gastrointestinal stromal tumors, which arise from the interstitial cells of Cajal, are spindle-cell neoplasms that should stain for c-kit (CD117).
胃肠道间质瘤(GIST),起源于Cajal间质细胞,是梭形细胞肿瘤,应进行c-kit(CD117)免疫组化染色。
Leiomyomas arise from smooth muscle cells. Leiomyoma is the second entity in the differential for are spindle-cell neoplasm. It stains for smooth muscle markers but not c-kit.
平滑肌瘤,起源于平滑肌细胞。平滑肌瘤是梭形细胞肿瘤的第二个鉴别诊断。它表达平滑肌标记物,但不表达CD117。
Carcinoids may be mucosal or submucosal; they have similar morphology to carcinoids elsewhere. Carcinoids may be sporadic, they may arise in multinodular form in response to autoimmune gastritis, or they may be associated with multiple endocrine neoplasia (MEN) syndromes.
类癌,可以位于粘膜内或粘膜下;其形态类似于其他部位。类癌可能是散发性,可能对自身免疫性胃炎的反应而呈现结节形式,也可能与多发性内分泌肿瘤(MEN)综合征有关。
十二指肠(Duodenum)
Duodenum is included here as duodenal tissue often accompanies stomach tissue in biopsy specimens, and the pathology in some cases is continuous. A duodenal biopsy may be performed because of combined gastritis and duodenitis, with or without peptic ulcer disease; to investigate suspected malabsorption syndromes, such as celiac disease; or to diagnose a mass lesion.
此处包括十二指肠,因为活检标本中十二指肠组织常伴随胃组织,并且,某些病例的病理是连续的。十二指肠活检的原因包括:组合性胃炎和十二指肠炎,伴或不伴消化性溃疡病;可疑的吸收不良综合征,如乳糜泻;或肿块病变的诊断。
Normal duodenal mucosa is characterized by narrow villi that project above the mucosal surface. The epithelium is intestinal type, which means goblet cells are interspersed among the absorptive cells. Lymphocytes, plasma cells, and eosinophils are normal inhabitants of the lamina propria. A few intraepithelial lymphocytes may be seen, although they should not be found at the tips of the villi. Under the muscularis mucosa are collections of mucous glands called Brunner’s glands, which stain bright pink on PAS/AB.
正常十二指肠粘膜的特征:狭窄的绒毛,突出在粘膜表面之上。上皮为肠型,即杯状细胞散在分布于吸收细胞之间。淋巴细胞、浆细胞和嗜酸性粒细胞是固有层的正常成分。可以看到少数上皮内淋巴细胞,但不应在绒毛顶端发现。粘膜肌层下方是一团粘液腺,称为Brunner腺(十二指肠腺),PAS/AB染色呈明亮的粉红色。
慢性消化性十二指肠炎(Chronic Peptic Duodenitis)
In severe gastritis, the inflammation and increased acid secretion may spill into the duodenum. In response to the lowered pH, the duodenum may “turn itself into stomach” or acquire gastric-type metaplasia. This shows up as metaplastic mucinous cells lining the villi (Figure 7.11) and is very obvious on PAS/AB stain because of the pink color of the gastric-type mucin. Other changes include Brunner’s gland hyperplasia, which is a mucosal (as opposed to a submucosal) proliferation of glands, and increased inflammation in the lamina propria. Advanced cases may ulcerate.
在严重的胃炎中,炎症和增加的酸分泌可能溢出到十二指肠。由于pH值降低,十二指肠可能“变成胃”或获得胃型化生。这表现为绒毛被覆化生性粘液细胞(图7.11),PAS/AB染色非常明显,因为胃型粘液呈粉红色。其他改变包括Brunner腺增生,这是腺体的粘膜部位(而不是粘膜下)增生,以及固有层的炎症增加。晚期病例可能形成溃疡。
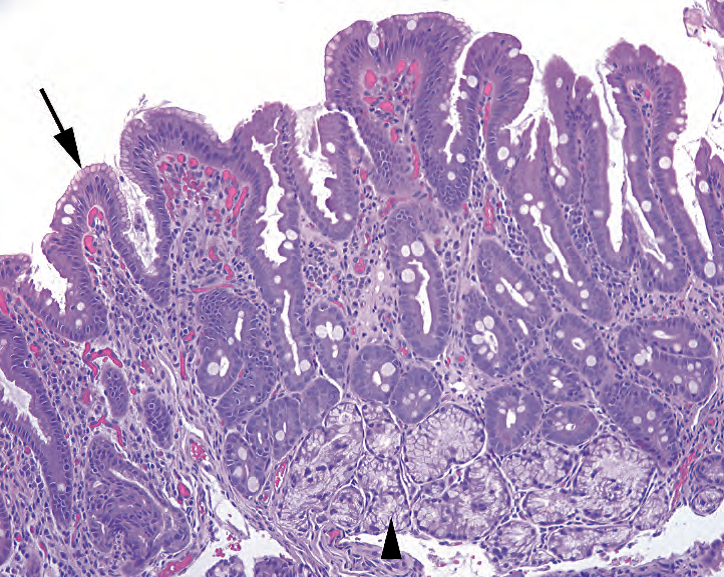
Figure 7.11. Chronic peptic duodenitis. At the surface, there is a subtle metaplastic change (arrow), where the normal absorptive and goblet cells are replaced by mucinous, foveolar-type cells, similar to those seen in antrum. There is increased chronic inflammation in the lamina propria and Brunner’s gland hyperplasia (arrowhead).
图7.11.慢性消化性十二指肠炎。表面有细微的化生改变(箭号),此处,正常的吸收细胞和杯状细胞被粘液性的小凹型细胞取代,类似于胃窦中看到的细胞。固有层慢性炎症细胞增多,Brunner腺增生(箭头)。
乳糜泻(Celiac Disease)
Although celiac disease, or sprue, is not very common, requests to rule it out are fairly frequent. The classic picture of advanced celiac disease is that of a completely flattened mucosa, with total loss of villi, such that the duodenum mimics colon (Figure 7.12). The absorptive epithelium loses its brush border and flattens into a low cuboidal layer, hence the resulting malabsorption. However, more subtle findings include villous blunting, or loss of villous height, and prominent intraepithelial lymphocytes at the tips of the remaining villi. Evaluating villous blunting can be difficult in a poorly oriented or mangled mucosal fragment; also keep in mind that the differential for villous atrophy is long and is only diagnostic of celiac disease if the serology and clinical picture agrees.
虽然乳糜泻,或口炎性腹泻,不是很常见,但临床经常要求排除它。晚期乳糜泻的典型表现是粘膜变得完全扁平,绒毛完全丧失,十二指肠貌似结肠(图7.12)。吸收上皮失去刷状缘,变扁平,成为一层低立方形细胞,因此导致吸收不良。然而,更细微的发现包括绒毛变钝,或变矮,以及在剩余绒毛的顶端有显著的上皮内淋巴细胞。在包埋方向不对或破损的粘膜碎屑中,可能难以评估绒毛变钝;还要记住,绒毛萎缩的鉴别是长期的过程,只有在血清学和临床表现相一致的情况下才诊断乳糜泻。
Infections of the duodenum include those caused by H. pylori, which can rarely occur in the setting of gastric mucin cell metaplasia, and giardiasis. Giardia is very sneaky, as the organisms hide in the luminal debris and do not cause inflammatory changes. In the immunocompromised, collections of foamy histiocytes stuffing the lamina propria may represent Mycobacterium avium-intracellulare infection. However, the differential for stuffed macrophages also includes Whipple’s disease, in which the macrophages are digesting Gram-positive rods and PAS-positive granules.
十二指肠的感染性疾病包括:HP引起的感染,罕见于胃粘液细胞化生和贾第鞭毛虫病的情况下。贾第虫非常隐匿,因为它们隐藏在管腔碎屑中,不会引起炎症改变。在免疫功能低下患者,充满固有层的泡沫状组织细胞聚集可能代表鸟分枝杆菌的细胞内感染。然而,填充巨噬细胞的鉴别诊断也包括Whipple病,即巨噬细胞正在消化革兰阳性杆菌和PAS阳性颗粒。
Neoplasms of the duodenum are unusual, but the most common of these are tubular adenomas (Figure 7.13), carcinoid tumors, and lymphoma (usually MALT type). Remember the intimate association of the duodenum to the pancreas and common bile duct; an adenocarcinoma found in duodenum may be originating in any of these organs.
十二指肠肿瘤少见,但最常见的是管状腺瘤(图7.13)、类癌和淋巴瘤(通常为MALT型)。记住十二指肠与胰腺和胆总管的紧密联系;十二指肠腺发现的腺癌可能起源于这些器官中的任何一个。
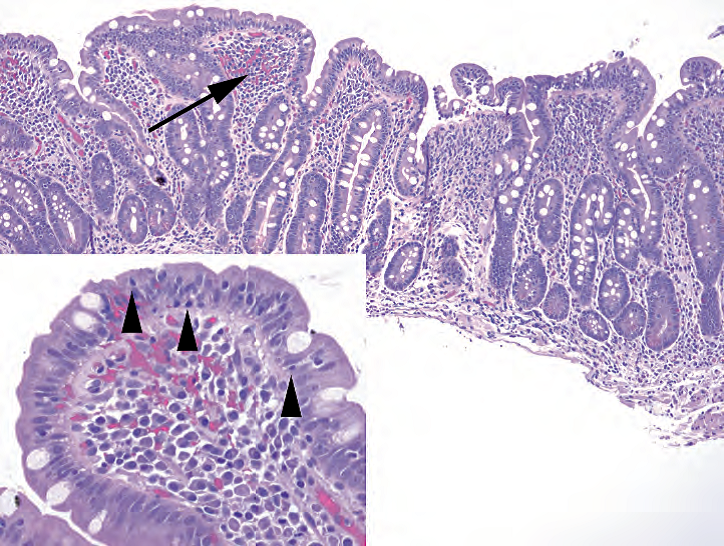
Figure 7.12. Celiac disease. The normal villi are blunted almost out of existence, with the duodenal mucosa resembling colon. There is chronic inflammation within the lamina propria (arrow). Inset: Increased numbers of intraepithelial lymphocytes are present (arrowheads).
图7.12.乳糜泻。正常绒毛变钝,几乎不存在,十二指肠粘膜类似结肠。固有层内有慢性炎症(箭号)。插图:上皮内淋巴细胞数量增加(箭头)。
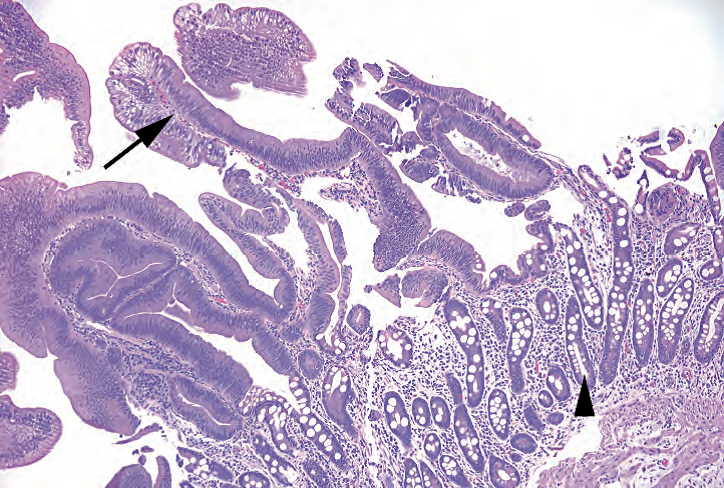
Figure 7.13. Duodenal adenoma. As in the colon, the tubular adenoma is characterized by low-grade dysplasia, showing crowded and elongated nuclei and loss of mucinous differentiation (arrow). Residual duodenal mucosa is seen underneath the adenoma (arrowhead).
图7.13.十二指肠腺瘤。正如结肠,管状腺瘤的特征是低级别异型增生,表现为细胞核密集和拉长,失去粘液分化(箭号)。腺瘤下方可见残留的十二指肠粘膜(箭头)。
来源:
The Practice of Surgical Pathology:A Beginner’s Guide to the Diagnostic Process
外科病理学实践:诊断过程的初学者指南
Diana Weedman Molavi, MD, PhD
Sinai Hospital, Baltimore, Maryland
ISBN: 978-0-387-74485-8 e-ISBN: 978-0-387-74486-5
Library of Congress Control Number: 2007932936
© 2008 Springer Science+Business Media, LLC
仅供学习交流,不得用于其他任何途径。如有侵权,请联系删除。
本站欢迎原创文章投稿,来稿一经采用稿酬从优,投稿邮箱tougao@ipathology.com.cn
相关阅读
 数据加载中
数据加载中
我要评论

热点导读
-

淋巴瘤诊断中CD30检测那些事(五)
强子 华夏病理2022-06-02 -
【以例学病】肺结节状淋巴组织增生
华夏病理 华夏病理2022-05-31 -

这不是演习-一例穿刺活检的艰难诊断路
强子 华夏病理2022-05-26 -

黏液性血性胸水一例技术处理及诊断经验分享
华夏病理 华夏病理2022-05-25 -

中老年女性,怎么突发喘气困难?低度恶性纤维/肌纤维母细胞性肉瘤一例
华夏病理 华夏病理2022-05-07







共0条评论